BK polyomavirus (BKPyV, historically BKV) is a ubiquitous human Betapolyomavirus with seroprevalence >80–90% in adults. Primary infection usually occurs in childhood, is asymptomatic or causes mild respiratory-like illness, and is followed by lifelong latency, particularly in renal tubular epithelial and uroepithelial cells.

In immunocompetent hosts, low-level viruria occurs in about 5–10% of adults. Clinically significant disease arises mainly when cellular immunity is impaired, especially after kidney transplantation (BK virus–associated nephropathy, ureteric stenosis) or hematopoietic stem cell transplantation (hemorrhagic cystitis).
Taxonomy and Classification
- Family: Polyomaviridae
- Genus: Betapolyomavirus
- Species: Human polyomavirus 1 (BK polyomavirus)
- Phylogenetic relationships:
- ~75% sequence homology with JC virus (JCPyV) and ~69% with simian virus 40 (SV40).
- Human polyomaviruses now include multiple additional species (e.g., KI, WU, Merkel cell polyomavirus), but BKPyV and JCPyV remain the most clinically important in transplantation.
- Genotype / subtype system:
- Four principal genotypes (I–IV) defined chiefly by VP1 gene sequence.
- Genotype I ≈ 80% of isolates globally; genotype IV ≈ 15%; genotypes II and III are rare.
- Geographical associations: subtype Ia in Africa, Ib-1 in Southeast Asia, Ib-2 in Europe, Ic in Northeast Asia.
- Some data indicate that genotypes II, III, IV and Ib1/Ib2 behave as distinct serotypes, with implications for neutralizing antibodies and immune escape.
Structure and Morphology
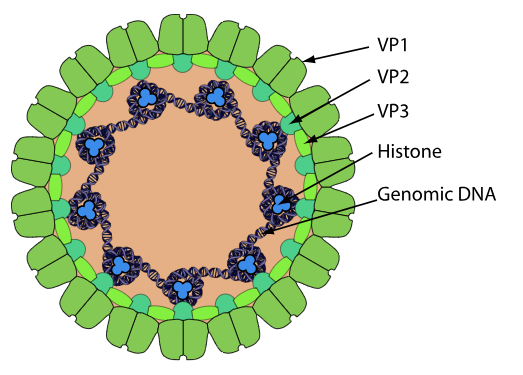
Figure: – Structure and Morphology of BK polyomavirus (Source: https://viralzone.expasy.org/148)
- Virion:
- Small, non-enveloped, icosahedral particle ~40–45 nm in diameter, highly stable and resistant to environmental stress.
- Capsid composed of 72 VP1 pentamers arranged with T=7 icosahedral symmetry, each associated internally with minor capsid proteins VP2 or VP3.
- Capsid proteins (structural):
VP1:
- Major capsid protein (~80% of virion protein mass) and principal target of neutralizing antibodies.
- Surface loops of VP1 determine receptor binding, tissue tropism, and are primary sites of genotype and antigenic variation.
VP2 / VP3:
- Minor structural proteins associated with VP1 pentamers on the inner surface; participate in genome encapsidation and cell entry.
- Cryo-EM reveals density consistent with ordered VP2/3 positioning and contacts with encapsidated DNA.
- Internal organization:
- Two concentric shells of electron density in cryo-EM attributed to ordered regions of the circular dsDNA genome and associated histone-like proteins.
Genome Organization and Proteins
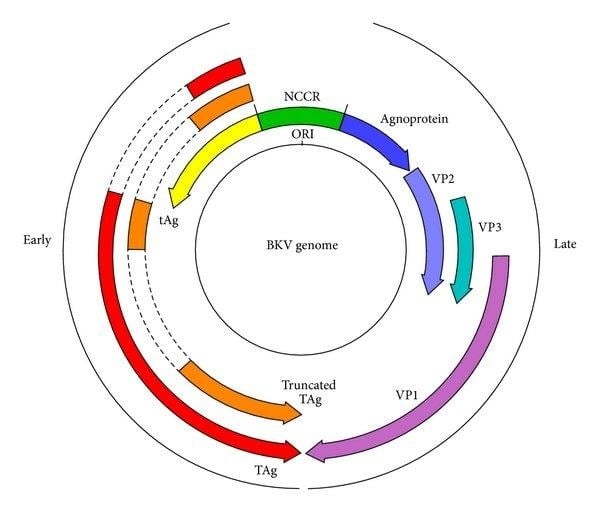
Figure: Schematic diagram illustrating the organization of the dsDNA genome of BK virus. The open reading frames are represented by arrows with alternative splicing events highlighted by dashed lines. The origin of replication (ORI) within the noncoding control region (NCCR), from which transcription of early and late mRNAs proceeds, is indicated. (Human Polyomavirus Reactivation: Disease Pathogenesis and Treatment Approaches – Scientific Figure on ResearchGate. Available from: https://www.researchgate.net/figure/Schematic-diagram-illustrating-the-organisation-of-the-dsDNA-genome-of-BK-virus-The-open_fig2_237057787 [accessed 2 Mar 2026])
- Genome:
Circular dsDNA ~5.1 kb partitioned into:
- Early region (regulatory proteins LT and st, plus splice variants)
- Late region (structural proteins VP1, VP2, VP3, agnoprotein)
- Non-coding control region (NCCR) containing origin of replication and transcriptional regulatory elements.
- Early proteins:
Large T antigen (LT):
- Essential for viral DNA replication via helicase and ATPase activity.
- Drives host cell into S-phase by binding and inactivating p53 and retinoblastoma (Rb) family proteins, promoting cell-cycle progression and persistent infection.
- Contains nuclear localization signals, origin-binding domains, and interaction motifs with cellular factors (e.g., p53, Rb, Hsc70).
Small t antigen (st):
- Modulates host signaling, including protein phosphatase 2A (PP2A), thereby influencing cell-cycle control and potentially oncogenic pathways.
- Late proteins:
- VP1 – major capsid protein as above; key for receptor engagement and serotyping.
- VP2 / VP3 – minor capsid proteins involved in entry, intracellular trafficking, and uncoating.
- Agnoprotein:
- Small accessory protein encoded by late region contributes to efficient virion assembly, release, and possibly modulation of host responses, though exact function remains incompletely defined.
- Regulatory elements:
NCCR:
- ~400 bp region with origin of replication and multiple transcription factors–binding sites controlling early and late gene expression.
- Archetype vs rearranged NCCRs: rearrangements (duplications/deletions) are strongly associated with enhanced replication, altered tropism, and high viral loads in transplant recipients with nephropathy and hemorrhagic cystitis.
Viral microRNA:
- BKPyV encodes miRNA (BKPyV-3p) targeting LT transcripts, down-regulating viral replication and potentially limiting antigen expression to evade immune detection.
Replication Cycle
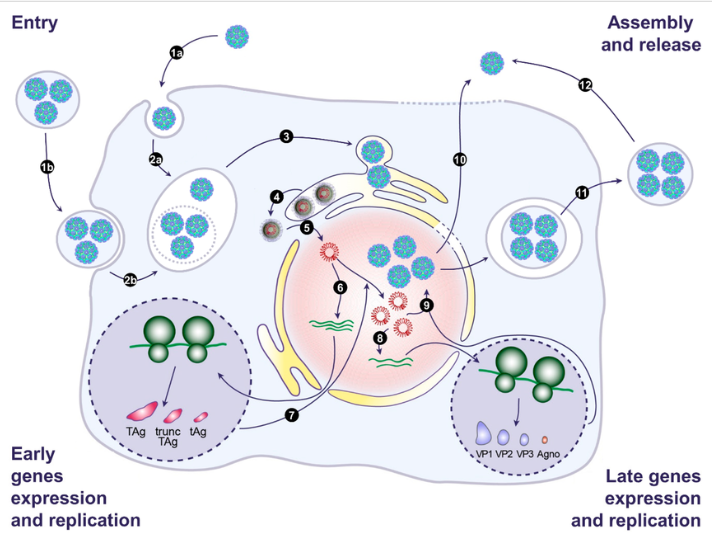
Figure: – Replication Cycle (DOI: 10.3390/v12101086)
Attachment and Entry
Receptor binding and cell targeting
- VP1–sialic acid interaction: VP1 surface loops recognize sialylated glycoconjugates on renal and urothelial cells; receptor specificity contributes to tropism.
- Genotype-specific VP1 variations modulate receptor affinity and may influence tissue tropism and immune escape.
Endocytic uptake
- Polyomaviruses use caveolin-dependent, clathrin-mediated, and/or other endocytic routes depending on cell type; BKPyV follows similar pathways defined mainly in kidney epithelial models and SV40 studies.
- Virions are internalized into endosomes and trafficked toward the endoplasmic reticulum (ER).
Intracellular Trafficking and Uncoating
Endosomal transport to ER
- After endocytosis, BKPyV traffics through the endosomal system to the ER, where key disassembly steps occur.
- pH-dependent and host chaperone-dependent conformational changes gradually destabilize the capsid.
ER-associated disassembly
- In the ER, polyomaviruses exploit ER chaperones and disulfide isomerases to rearrange VP1 disulfide bonds and expose internal VP2/VP3 and the genome.
- Partial uncoating yields a subviral particle containing VP1, VP2/3, and the minichromosome (viral DNA wrapped in host histones).
Nuclear Import
Nuclear localization signals (NLS)
- VP2/VP3 and large T antigen (LT) harbor NLS motifs that recruit importins and mediate transport of the subviral complex across nuclear pores into the nucleus.
Establishment of the minichromosome
- In the nucleus, the circular ~5.1 kb dsDNA associates tightly with cellular histones to form a viral minichromosome, configuring the genome for transcription and replication.
Early Gene Expression and Cell-Cycle Manipulation
- Early transcription from NCCR
- Functions of early proteins
- Large T antigen (LT):
- Binds the viral origin of replication and forms multimeric helicase complexes to unwind DNA.
- Drives host cells into S phase and re-replication by binding and inactivating Rb family proteins (Rb, p107, p130) and modulating p53, thus bypassing normal cell-cycle checkpoints.
- Single-cell studies show that robust LT expression actually requires host DNA synthesis first; BKPyV preferentially amplifies in cells that have already entered S phase and then promotes S-phase re-entry from G2 (re-replication), leading to >4N host DNA content.
- Small t antigen (st):
- Interferes with host protein phosphatase 2A (PP2A) and signaling pathways, further supporting cell-cycle progression and potentially transformation.
- Large T antigen (LT):
- Dependence on host S phase and ribosome state
- BKPyV production is critically dependent on the host cell’s initial cell-cycle status; depletion of ribosomal protein S25 arrests cells in G0/G1 and G2/M and markedly reduces productive infection, showing that cell-cycle entry is a key early requirement.
- Overall, BKPyV relies on a pre-existing S phase, then extends and recycles S phase to maintain a replication-permissive environment.
Viral DNA Replication and DNA Damage Response (DDR)
Initiation of viral DNA replication
- LT binds the origin within the NCCR and recruits host DNA replication factors (MCM helicase, DNA polymerase α-primase, ligase, replication factor C) to commence bidirectional theta-type replication of the circular genome.
- Recent iPOND-MS work in murine polyomavirus confirms dense recruitment of host replisome components to newly synthesized viral DNA, consistent with similar mechanisms in BKPyV.
Activation of DNA damage response
- Viral DNA replication, not input virus or LT alone, is the main trigger for activating ATM- and ATR-mediated DDR pathways.
- BKPyV upregulates the mismatch repair (MMR) complex MutSα (Msh2/Msh6), which is required for full ATR pathway activation and for maintaining S-phase arrest favorable to replication.
- Inhibition of Msh6 leads to severe host DNA damage, impaired DDR activation, premature mitotic entry, and production of fewer, defective virions lacking VP2.
Host re-replication and nuclear enlargement
- DDR activation and LT together promote host DNA re-replication (S-phase re-entry from G2), causing polyploid nuclei—an in vitro phenotype that mirrors giant TAg-positive nuclei in kidney biopsies from patients with uncontrolled BKPyV replication.
Late Gene Expression, Assembly, and Maturation
Viron Release and Shedding
- Switch from early to late transcription
- Accumulated LT and changes in NCCR occupancy favor late gene transcription, producing mRNAs for VP1, VP2, VP3, and agnoprotein.
- NCCR rearrangements (duplications/deletions) can markedly increase late promoter activity and replication capacity, a process observed during persistent infection and reactivation.
- Capsid assembly in the nucleus
- VP1 pentamers assemble and associate with VP2/3 to form 72-pentamer icosahedral capsids; virion assembly occurs within intranuclear “replication centers” where newly synthesized genomes concentrate.
- Cryo-EM and urinary cell studies show two concentric shells of density attributed to ordered viral DNA and VP2/3 beneath the VP1 shell, suggesting highly coordinated genome–capsid interactions in assembly.
- Genome packaging
- Replicated minichromosomes (viral DNA plus histones) are inserted into pre-assembled VP1/VP2/3 capsids; efficient encapsidation depends on intact late proteins and DDR-maintained nuclear integrity.
Virion Release and Shedding
- Cell lysis
- The classical pathway is lytic cell death, dominated by necrosis rather than apoptosis, leading to high-titer release of virions into the tubular lumen and urine (viruria) and then into peritubular capillaries (viremia).
- Extracellular vesicle (EV)–mediated release
- Recent work shows BKPyV is also released enclosed within extracellular vesicles, which may:
- Permit non-lytic egress.
- Shield virions from neutralizing antibodies.
- Modify cell tropism and immune sensing.
- Recent work shows BKPyV is also released enclosed within extracellular vesicles, which may:
- Integration and recombination during persistence
- During persistent infection and reactivation, BKPyV genomes undergo extensive recombination, especially in the NCCR, generating variants with enhanced replication.
- Rarely, chromosomal integration of viral DNA occurs and may contribute to high-level replication and possibly oncogenesis, though this remains uncommon in routine infection.
Pathogenesis and Host Immune Response
- Persistence and latency:
- After primary infection, BKPyV establishes low-level persistent infection in the genitourinary tract and possibly other tissues (e.g., tonsils, brain, endothelial cells).
- Cellular immune surveillance, particularly CD8+ T cells, maintains latency and restricts replication in immunocompetent hosts.
- Reactivation triggers:
- Pharmacological immunosuppression (e.g., calcineurin inhibitors, antimetabolites, steroids), HIV infection, and other states of impaired cellular immunity promote viral reactivation with high-level viruria and viremia.
- In kidney transplantation, over-immunosuppression, prior rejection episodes, HLA mismatching, older age, and ureteral stenting are important risk factors.
- Immune responses:
- Humoral immunity:
- High seroprevalence; BKPyV-specific IgG directed mainly against VP1 and capable of neutralizing infection in vitro.
- However, prior seropositivity does not reliably protect against reactivation or nephropathy in transplant recipients, partly due to genotype/serotype variation and immune evasion.
- Cellular immunity:
- BKPyV-specific CD4+ and CD8+ T cells recognizing LT, VP1 and other epitopes are critical for control; robust T-cell responses correlate with viral clearance and protection from nephropathy.
- Impaired BK-specific T-cell responses (quantitatively and qualitatively) are strongly associated with persistent viremia and disease.
- Immune evasion:
- Down-modulation of LT expression via viral miRNA; possible alteration of antigenic epitopes via VP1 and NCCR mutations; latency in immune-privileged or low-surveillance sites.
- Release of virions within extracellular vesicles may shield them from neutralizing antibodies.
- Humoral immunity:
- Tissue damage:
- Productive lytic infection of renal tubular epithelial cells causes cytopathic changes, tubular injury, and interstitial inflammation that can progress to BKPyV-associated nephropathy (BKVAN) with fibrosis and allograft failure.
- In hematopoietic stem cell transplantation, massive viral replication in bladder urothelium leads to hemorrhagic cystitis, with urothelial necrosis, hematuria, and pain.
Epidemiology and Transmission
- Seroprevalence:
- Infection acquired predominantly in early childhood, with >80–90% of adults seropositive worldwide.
- Asymptomatic viruria in 5–10% of healthy adults; viral loads low (typically <3 log10 copies/mL).
- Transmission routes:
- Likely respiratory and/or fecal-oral transmission during childhood; BK DNA detected in nasopharyngeal aspirates of children with respiratory infections.
- Vertical transmission and transmission via blood products, semen, and organ transplantation, particularly renal allografts, have been described.
- High-risk populations:
Kidney transplant recipients:
- BK viremia in ~10–20% of recipients, with BKVAN in up to 10% depending on immunosuppression and screening protocols.
- BK viremia is now a leading cause of early graft dysfunction and loss.
Allogeneic hematopoietic stem cell transplant (HSCT) recipients:
- Viruria in 60–80% and BK-associated hemorrhagic cystitis in 4–27% of pediatric cohorts, with significant morbidity.
Other settings:
- Rare cases of pneumonitis, retinitis, meningoencephalitis, and possible associations with malignancies (e.g., urothelial carcinoma, prostate cancer), though causal links remain controversial.
Clinical Manifestations
In immunocompetent hosts:
- Most primary infections are asymptomatic or mild, non-specific upper respiratory or flu-like syndromes in childhood.
- Persistent low-level viruria without renal dysfunction.
In kidney transplantation:
- BK viremia and viruria precede tissue-invasive disease; progressive rise in plasma viral load strongly predicts BKVAN.
- BKPyV-associated nephropathy:
- Clinical features: rising serum creatinine, reduced graft function, often without specific urinary symptoms.
- Histology: tubulointerstitial nephritis with viral inclusions, “decoy cells,” positive immunohistochemistry for LT antigen, and interstitial fibrosis in advanced stages.
Ureteric stenosis: due to viral-induced urothelial damage and fibrosis, leading to obstructive uropathy.
- In HSCT:
Hemorrhagic cystitis:
- Manifestations: dysuria, frequency, bladder pain, hematuria ranging from microscopic to life-threatening gross hematuria with clot retention.
- Onset typically after engraftment and associated with high-level viruria and viremia.
- Disease burden: prolonged hospitalization, need for intensive supportive care, and potential renal compromise.
Other reported manifestations:
- Encephalitis, meningitis, pneumonitis, retinitis, and possible associations with tumors (urothelial carcinoma, prostate and renal cell carcinoma) have been reported, but evidence for causality is limited and confounded by co-morbidities and immunosuppression.
Laboratory Diagnosis
- Direct detection:
Quantitative PCR (qPCR) for BK DNA in urine and plasma is the mainstay of diagnosis and monitoring.
- Urine PCR: highly sensitive for reactivation (viruria often precedes viremia) but less specific for disease; useful for screening.
- Plasma PCR: correlates more closely with tissue-invasive disease; persistent high-level viremia is a strong predictor of BKVAN and severe hemorrhagic cystitis.
- Issues: inter-assay variability and lack of full standardization; movement toward reporting in international units is ongoing.
Decoy cells:
- Urine cytology identifies enlarged epithelial cells with intranuclear viral inclusions; sensitive but not specific for BKV (JC virus can also cause these changes).
Histopathology:
- Renal biopsy remains the gold standard for diagnosing BKVAN, showing characteristic viral cytopathic changes and confirmed by immunohistochemistry for LT antigen or in situ hybridization for viral DNA.
- Genotyping and viral diversity analysis:
- VP1 PCR and sequencing used for genotype and subtype determination; useful for epidemiological studies, tracking transmission, and understanding potential genotype–disease associations.
- NCCR sequencing identifies rearrangements associated with high replication competence and severe disease.
- Serology:
- VP1-based ELISAs and neutralization assays measure BK-specific antibodies, but lack of standardized assays and cross-reactivity limit routine clinical use.
- Immune monitoring:
- ELISPOT and flow-cytometric assays quantifying BK-specific T-cell responses are promising tools to stratify risk and guide immunosuppression adjustment, but are not yet widely standardized.
Treatment and Antiviral Therapy
Cornerstone: modulation of immunosuppression
- Stepwise reduction of calcineurin inhibitors and/or antimetabolites is the primary, evidence-based intervention for BKVAN and high-grade viremia, leading to viral load reduction in many patients.
- Trade-off: increased risk of acute rejection, requiring careful individual balancing and close monitoring.
Antiviral agents (off-label, limited evidence):
- Cidofovir:
- Nucleotide analogue with in vitro anti-BK activity; small, uncontrolled series suggest possible benefit, but nephrotoxicity limits use and randomized data are lacking.
- Leflunomide:
- Immunomodulatory agent with reported anti-BK activity; observational studies show mixed results; drug levels and hepatotoxicity require monitoring.
- Fluoroquinolones and brincidofovir have been explored with inconclusive or modest effects; none are approved specifically for BKPyV.
Immune-based therapies:
BK-specific T-cell therapy:
- Adoptive transfer of virus-specific T cells (VSTs) has shown encouraging responses in refractory BKV disease (particularly hemorrhagic cystitis) in small series; still experimental but a key emerging strategy.
Intravenous immunoglobulin (IVIg):
- Contains polyclonal anti-BK antibodies; sometimes used as adjunctive therapy, but evidence for efficacy is limited.
Supportive measures:
- For hemorrhagic cystitis: aggressive hydration, bladder irrigation, analgesia, management of clot retention, and transfusion support as needed.
Prevention and Control
- In kidney transplantation:
Screening protocols:
- Regular BK viremia monitoring (e.g., monthly for first 6–9 months, then less frequently up to 2 years) enables pre-emptive immunosuppression reduction before irreversible nephropathy develops.
Immunosuppression strategies:
- Use of less intensive regimens, careful minimization after early rejection risk period, and avoidance of excessive combined immunosuppression reduce BK risk.
Graft and donor factors:
- Consideration of donor and recipient BK serostatus, cold ischemia time, and judicious use of ureteral stents may modulate risk, though data remain heterogeneous.
In HSCT:
- No standardized, universally accepted prevention strategy; some centers monitor viruria and viremia and adjust conditioning or immunosuppression, but evidence is variable.
- Optimization of conditioning regimens, timely management of graft-versus-host disease, and avoidance of prolonged profound lymphopenia may reduce risk.
Vaccines:
- No licensed BKPyV vaccine; conceptually attractive given stable capsid antigens and high disease burden in transplantation, but at present only hypothetical and pre-clinical.
Infection control:
- Given high background prevalence and presumed common childhood acquisition, standard precautions suffice; no specific isolation strategies are recommended beyond usual transplant infection-control practices.
Conclusion
BK polyomavirus is a small, structurally simple but clinically significant human polyomavirus whose pathogenic potential emerges primarily in the setting of impaired cellular immunity. Detailed understanding of its genome organization, NCCR variability, and immune interactions has clarified why reactivation leads to nephropathy and hemorrhagic cystitis in transplant recipients while remaining silent in most others. Diagnosis relies on standardized nucleic acid testing combined with histopathology, and current management remains centered on carefully calibrated reduction of immunosuppression, supported by limited antiviral and emerging immune-based approaches. Persistent gaps include the absence of approved antivirals or vaccines, incomplete standardization of diagnostics and immune monitoring, and uncertain links to malignancy and extra-renal disease.
References
- Human Polyomavirus Reactivation: Disease Pathogenesis and Treatment Approaches – Scientific Figure on ResearchGate. Available from: https://www.researchgate.net/figure/Schematic-diagram-illustrating-the-organisation-of-the-dsDNA-genome-of-BK-virus-The-open_fig2_237057787 [accessed 2 Mar 2026]
- Ambalathingal, G. R., Francis, R. S., Smyth, M. J., Smith, C., & Khanna, R. (2017). BK Polyomavirus: clinical aspects, immune regulation, and emerging therapies. Clinical Microbiology Reviews, 30(2), 503–528. https://doi.org/10.1128/cmr.00074-16
- Bennett, S. M., Broekema, N. M., & Imperiale, M. J. (2012). BK polyomavirus: emerging pathogen. Microbes and Infection, 14(9), 672–683. https://doi.org/10.1016/j.micinf.2012.02.002
- Furmaga, J., Kowalczyk, M., Zapolski, T., Furmaga, O., Krakowski, L., Rudzki, G., Jaroszyński, A., & Jakubczak, A. (2021). BK Polyomavirus—Biology, Genomic Variation and Diagnosis. Viruses, 13(8), 1502. https://doi.org/10.3390/v13081502
- Helle, F., Brochot, E., Handala, L., Martin, E., Castelain, S., Francois, C., & Duverlie, G. (2017). Biology of the BKPYV: An update. Viruses, 9(11), 327. https://doi.org/10.3390/v9110327
- Hirsch, H. H., & Snydman, D. R. (2005). BK Virus: Opportunity makes a pathogen. Clinical Infectious Diseases, 41(3), 354–360. https://doi.org/10.1086/431488
- Kant, S., Dasgupta, A., Bagnasco, S., & Brennan, D. C. (2022). BK Virus Nephropathy in Kidney Transplantation: A State-of-the-Art Review. Viruses, 14(8), 1616. https://doi.org/10.3390/v14081616
- Peras, M., Bilić, E., & Mareković, I. (2025). Recent Insights into the Pathogenesis, Diagnostics, and Treatment of BK Virus Infections in Children After Hematopoietic Stem Cell Transplantation. Pathogens, 14(3), 236. https://doi.org/10.3390/pathogens14030236
- Sawinski, D., & Goral, S. (2014). BK virus infection: an update on diagnosis and treatment. Nephrology Dialysis Transplantation, 30(2), 209–217. https://doi.org/10.1093/ndt/gfu023
- Trang, V. D., Rockett, R., Jeoffreys, N., Trung, N. V., An, H. P. H., Kok, J., & Dwyer, D. E. (2017). BK Polyomavirus: A Review of the virology, pathogenesis, Clinical and Laboratory features, and Treatment. Future Virology, 12(8), 439–459. https://doi.org/10.2217/fvl-2017-0013
- Blackard, J. T., Davies, S. M., & Laskin, B. L. (2020). BK polyomavirus diversity—Why viral variation matters. Reviews in Medical Virology, 30(4), e2102. https://doi.org/10.1002/rmv.2102
- Helle, F., Handala, L., Bentz, M., Duverlie, G., & Brochot, E. (2020). Intercellular Transmission of Naked Viruses through Extracellular Vesicles: Focus on Polyomaviruses. Viruses, 12(10), 1086. https://doi.org/10.3390/v12101086
