Corynebacterium diphtheriae, also known as Kleb’s Loeffler bacterium, is responsible for causing a serious infection known as diphtheria which spreads from human to human mainly via respiratory droplets while sneezing or coughing.
This bacterium is responsible for the production of toxins which attach to the tissues of the respiratory system and cause symptoms ranging from sore throat, fever, swollen neck glands and weakness.
Taxonomy and Classification
Domain: Bacteria
Kingdom: Bacillati
Phylum: Actinomycetota
Class: Actinomycetes
Order: Mycobacteriales
Family: Corynebacteriaceae
Genus: Corynebacterium
Species: C. diphtheria
Morphology and Microscopy
- Gram positive, club-shaped bacillus arranged either in pair or small groups or in clusters
- Approximately 3-5µm * 0.5-0.8µm in diameter
- Non-motile
- Non-capsulated
- Non-sporing
- Presence of pili
Under microscope, they appear as purple colored bacillus arranged in Chinese letter like or V or L shaped arrangement. Appears green in Albert’s stain along with bluish black metachromatic granules at the end.
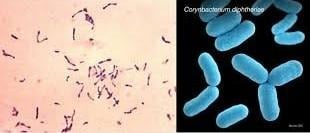
Figure 1: Structure of C. diphtheria
Source: https://microbenotes.com/corynebacterium-diphtheriae/
Cultural and Growth Characteristics
- Aerobes and facultative anaerobes
- Optimum temperature: 37˚C
- Optimum pH: 7.2
- Show minimal growth on ordinary media
- Media enriched with egg, blood or serum is necessary for the growth of this organism
- On Blood Agar: small, granular, irregular margins, grey in color, presence or absence of hemolysis.
- Loeffler’s Serum Slope: circular, white, opaque, yellowish tint and shows rapid growth.
- Tellurite Blood Agar: brown to black producing brown-black halo region.
- Tinsdale’s Medium: grayish black colonies having dark brown halo.
Biotypes of Corynebacterium diphtheria
C. diphtheriae is classified into 4 biotypes based on the colony characteristics on Tellurite Medium and other properties:
- Gravis
- Are short rods with few granules and show some degree of pleomorphism.
- On Tellurite Medium it shows daisy head colonies with flat colonies having raised dark center and created margin and radial striations.
- It is variable hemolysis.
- It is positive for glycogen or starch fermentation.
- Intermedius
- Are long barred in form with poor granulation and show pleomorphism.
- On Tellurite Medium, a frog’s egg-like colony is formed which has a dull granular center with glistening periphery and light ring near the edge.
- Shows non-hemolytic colonies.
- It is negative for glycogen or starch fermentation.
- Mitis
- Are long curved in shape with prominent granules and show pleomorphism.
- On Tellurite Medium, Poached Egg like colony is formed having shiny, flat with central elevation
- Shows hemolytic colonies.
- It is negative for glycogen or starch fermentation.
- Belfanti
- There are less known biotypes of C. diphtheria.
Biochemical and Identification Tests
| Tests | Results |
| Gram Staining | Positive |
| Catalase | Positive |
| Oxidase | Negative |
| Indole | Negative |
| OF (Oxidative-Fermentative) | Fermentative |
| MR (Methyl Red) | Positive |
| H2S | Positive |
| Urease | Negative |
| Gelatin Hydrolysis | Negative |
| Nitrate Reduction | Positive |
| Gas | Variable |
| Coagulase | Negative |
| Citrate | Negative |
| Cetrimide | Negative |
| CAMP | Negative |
| Fermentation of | |
| Glucose | Positive |
| Maltose | Positive |
| Dextrose | Positive |
| Lactose | Negative |
| Mannitol | Negative |
| Fructose | Positive |
| Arabinose | Positive |
| Mannose | Positive |
| Xylose | Negative |
| Sucrose | Negative |
| Mannose | Positive |
| Galactose | Positive |
| Starch | Negative |
| Trehalose | Negative |
| Glycogen | Negative |
| Enzymatic Reactions | |
| Esculin Hydrolysis | Negative |
| Alkaline Phosphatase | Negative |
| Casein Hydrolysis | Negative |
| Tellurite | Positive |
| Tyrosine Hydrolysis | Negative |
| Ornithine Decarboxylase | Negative |
Pathogenesis and Virulence Factors
- The source of infection by C. diphtheriae is through airborne droplets.
- The bacteria enters the body through the upper respiratory tract.
- After entry, they grow on the mucous membrane where they multiply and start producing toxins.
- This toxin then inhibits the synthesis of cellular protein which is responsible for the destruction of local tissue and development of pseudomembrane which is the characteristic feature of the disease.
- After that the toxin produced is absorbed through the blood stream which is then dispersed to the various parts of the body.
- This toxin causes various complications like polyneuropathies, nephritis, myocarditis and even thrombocytopenia.
The major virulence factor produced by C. diphtheriae is explained below:
- Diphtheria Toxin
- It is synthesized in the form of a precursor of molecular weight 58,700 Da having a polypeptide made up of 535 amino acids.
- It has two fragments : A which is active and B which is responsible for binding and has a molecular weight of 21,500 and 37,200 Da respectively.
- Fragment A is an active fragment that enters into the cell and then causes ADP ribosylation of elongation factor 2 which then causes inhibition of elongation factor 2 leading to inhibition of translation of synthesis of protein.
- Fragment B is responsible for binding to the host cell receptors which then helps in the entry.
- Adhesins
- Adhesins are pili, fimbriae, surface proteins that help in the attachment or adherence.
- Helps in the colonization in the upper respiratory tract.
- Iron Acquisition Systems
- Siderophores and other iron-binding proteins permit the survival of bacteria in iron-restricted environments.
- Production of diphtheria toxin is controlled by the level of iron, low iron level contributes to increased expression of toxin.
- Capsule
- Some strains are responsible for the production of polysaccharide capsules which withstand phagocytosis and assist in the evasion of the immune system.
Epidemiology and Transmission
Around the year 1920s, approximately 100,000-200,000 diphtheria cases were reported among which 13,000-15,000 deaths were reported each year. Then after the discovery of a vaccine against diphtheria in the 1940s, the cases decreased to around 19,000 in 1945 which is approximately 15 cases per 100,000 populations. In the late 1940s, after the discovery of diphtheria toxoid containing vaccines developed for the universal childhood vaccination program, there was a drastic drop in the case. In the United States, there were 14 reported cases of diphtheria from 1996-2018 which is on average less than 1 per year. There was a death of a 63 year old man who was returning from a country having endemic diphtheria to the United States. Death was observed in 1992 where a man of age 41 had multiple co-morbid conditions. In Russia, at the end of 1994, around 80,000 cases and approximately 2,000 deaths were recorded. In Singapore in 2008-2010, the survey showed that all the children below 17 years contain antitoxin immunity against diphtheria. Singapore is the place which is considered to be free from diphtheria as an outcome of childhood immunization programmes. In Europe, Eastern Europe and the United States, diphtheria outbreaks have occurred most among drug users and alcohol consumers. Diphtheria outbreaks are always associated with an immune carrier who returns from the region which is endemic of diphtheria. However, recent outbreaks in the United States and Europe are associated with visitors returning from Eastern Europe and Russia.
C. diphtheriae is transmitted via various ways which are explained below:
- Is transmitted through person-to-person contact.
- Transmitted via the droplets from the respiratory tract while sneezing and coughing.
- Spread through the contact with surfaces contaminated with the infected person’s respiratory droplet.
- Spread through direct contact with open sores or ulcers caused by C. diphtheriae.
Clinical Manifestations
The incubation period of infection by C. diphtheriae is usually 2-5 days, ranging from 1-10 days. There are various infections related to C. diphtheriae which are described below:
- Facial Diphtheria
- Involves infection of the posterior region of mouth, tonsils and proximal pharynx.
Symptoms are:
- Low grade fever
- Sore throat
- Malaise
- Formation of membrane on tonsils
- Anterior Nasal Diphtheria
- Involves the ulceration of the external nostrils and upper lip in infants.
Symptoms include:
- Mucoid or blood containing nasal discharge
- Laryngeal Diphtheria
- Occurs as a result of development of membrane especially on tonsils.
Symptoms are:
- Hoarseness in voice
- Dyspnea (difficulty in breathing)
- Brassy cough
- Death within week if remains untreated
- Systemic Complications
Occur due to diphtheria toxin which cause:
- Myocarditis
- Neuropathy involving cranial nerve, oculomotor and facial nerves
- Cutaneous Infections
Contains symptoms such as:
- Pain
- Redness
- Swelling
- Chronic non-healing ulcers with dirty grey membrane
- Punched-out ulcers with a membrane and edematous (blood containing) rolled borders.
- Infection on other sites
Other body parts such as the eye, ear, genitalia, vulva and vagina are infected with C. diphtheriae.
Laboratory Diagnosis
- Sample Collection and Transportation
- Specimens are swabs collected from the throat, nose and pieces of pseudo-membrane.
- Two swab samples are collected, one for direct smear and other for culture.
- Samples are collected prior to antibiotic treatment.
- Transfer is carried out in sterile empty containers or in silica gel sachets for immediate processing.
- Microscopy
- It appears as purple colored rods with Chinese letter-like arrangement under microscope.
- Cells contain metachromatic granules which are stained bluish-purple with methylene blue.
- Culture
- Specimens are inoculated on selective media such as Tellurite medium, enriched media like Loeffler’s Medium, Hoyle Medium or Cysteine-tellurite Blood Agar (CTBA) and non-selective media such as blood agar.
- On Blood Agar: Non-hemolytic colonies
- On Tellurite Medium: black to grayish black colonies are formed after incubation of 48 hours.
- On Loeffler’s Medium: Colonies are small, circular, white and opaque after incubation at 37˚C for 4-6 hours.
- On CTBA: brown halo is produced around colonies due to degradation of cysteine.
- Biochemical Tests
After culture, colonies from incubated plates are performed for biochemical test and are confirmed as C. diphtheria based on following test:
| Tests | Results |
| Gram Staining | Positive |
| Catalase | Positive |
| Oxidase | Negative |
| Indole | Negative |
| OF (Oxidative-Fermentative) | Fermentative |
| MR (Methyl Red) | Positive |
| H2S | Positive |
| Urease | Negative |
| Gelatin Hydrolysis | Negative |
| Nitrate Reduction | Positive |
| Gas | Variable |
| Coagulase | Negative |
| Citrate | Negative |
| Cetrimide | Negative |
| CAMP | Negative |
- Toxigenicity Test
- All strains should be tested for toxin production.
- Elek (Elek-Ouchterlony Immunodiffusion ) test is an in-vitro immunodiffusion test used for the detection of diphtheria toxin.
- Polymerase Chain Reaction based nucleic acid amplification method is used for the detection of tox genes in clinical samples.
- ELISA can be used for the detection of diphtheria toxin from clinical isolates.
- Immunochromatographic Strip detects toxins of C. diphtheriae within an hour.
- Tissue Culture
- Tissue culture test is performed for the detection of production of toxin in many eukaryotic cell lines such as Chinese hamster Ovary, African Green Monkey Kidney.
Treatments
- Erythromycin either orally or through injection is given for 14 days (40mg/kg per day with a maximum of 2 g/d).
- Procaine penicillin G is given intramuscularly for 14 days (300,000 U/d for patients having weight ˂10kg and 600,000 U/d for patients having weight ˃10kg).
- Patients showing allergies to Penicillin G or Erythromycin can be given Rifampin or Clindamycin.
Vaccination
Diphtheria Toxoid-containing Vaccines are explained below:
- DTaP
- Doses of 3 are given at an age of 2, 4 and 6 months.
- The interval between two doses is 4-8 weeks.
- Booster doses are given at age of 15 to 18 months and 4 to 6 years.
- Minimum interval between dose 3 and 4 is 6 months and minimum age is 12 months.
- If the 4th dose is given on or after the 4th birthday, the 5th dose is optional.
- Tdap
- First dose is given at age through 11 to 18 for adolescents who have completed DTaP vaccine schedule.
- For all persons a booster dose is given every 10 years.
Prevention and Control
The most effective prevention is vaccination however other preventive measures are explained below:
- Regular washing of hands using soap especially before eating and after using the toilet and closing mouth after coughing or sneezing.
- Avoid sharing food, drinks or utensils with a person infected with C. diphtheriae.
- Practicing good personal hygiene all the time.
- Not touching the face, eye, nose or mouth with unwashed or dirty hands.
- Wear a mask when infected and immediately visit a doctor for the treatment.
Conclusion
Corynebacterium diphtheriae is defined as Gram-positive, non-motile bacilli which most commonly spread via direct human to human contact or through airborne droplets which is responsible for causing serious infectious disease like breathing and swallowing problems and can cause sores on skin because of the production of toxin. The persons who are at risk with infection by C. diphtheriae are those who have partial vaccination or unvaccinated, those with weak immune systems and those who travel or live in areas having an outbreak of C. diphtheriae. It can be treated with medications and vaccination but in adverse conditions can damage kidneys, heart and nervous system.
References
- Baron, S. (Ed.). (1996). Corynebacterium diphtheriae. In Medical microbiology (4th ed.). University of Texas Medical Branch. https://www.ncbi.nlm.nih.gov/books/NBK7971/
- BrainKart. (n.d.). Corynebacterium diphtheriae: Morphology, cultural characteristics, pathogenicity, clinical manifestations, laboratory diagnosis, prophylaxis, treatment. BrainKart. https://www.brainkart.com/article/Corynebacterium-Diphtheriae_41014/
- Centers for Disease Control and Prevention. (2024, April 23). Chapter 7: Diphtheria. In Epidemiology and Prevention of Vaccine-Preventable Diseases (14th ed.). U.S. 4. Department of Health and Human Services. https://www.cdc.gov/pinkbook/hcp/table-of-contents/chapter-7-diphtheria.html
- Murphy, J. R. (1996). Corynebacterium diphtheriae. In S. Baron (Ed.), Medical microbiology (4th ed., Chap. 32). University of Texas Medical Branch at Galveston. https://www.ncbi.nlm.nih.gov/books/NBK7971/
- University of Basrah, Faculty of Science. (2023). Corynebacterium diphtheriae [PDF]. University of Basrah, Basrah, Iraq. https://faculty.uobasrah.edu.iq/uploads/teaching/1702970894.pdf
- Centers for Disease Control and Prevention. (2024). Chapter 7: Diphtheria. In Epidemiology and Prevention of Vaccine-Preventable Diseases (14th ed.). U.S. Department of Health and Human Services. https://www.cdc.gov/pinkbook/hcp/table-of-contents/chapter-7-diphtheria.html
- National Centre for Infectious Diseases. (n.d.). Diphtheria. https://www.ncid.sg/Health-Professionals/Diseases-and-Conditions/Pages/Diphtheria.aspx
- Communicable Diseases Agency. (n.d.). Diphtheria. https://www.cda.gov.sg/public/diseases/diphtheria/
