Coxiella burnetii is a small, gram negative obligate intracellular bacterium that is the causative agent of Q-fever, a zoonotic disease that affects both humans and animals. The organism infects domestic ruminants such as sheep, goats and cattle which acts as the main reservoirs of infection.
Infected animals often shed the bacteria in birth products, milk, urine and feces, contaminating the surrounding environment. Human infection most commonly occurs through inhalation of contaminated aerosols or dust originating from infected animals or their products. The bacterium is highly infectious and capable of surviving for long periods in harsh environmental conditions, which contributes to its wide global distribution. Due to its zoonotic nature, environmental persistence and occupational risk among livestock workers and veterinarians, C. burnetii remains an important pathogen of public health concern.
Classification of Coxiella burnetiid
Domain: Bacteria
Phylum: Protobacteria
Class: Gammaproteobacteria
Order: Legionellales
Family: Coxiellaceae
Genus: Coxiella
Species: Coxiella burnetii
https://www.sciencephoto.com/media/612285/view/coxiella-burnetti-bacteria-tem
Coxiella has been closely associated with members of Rickettsiae based on the comparisons of their phenotypic characteristics. Later, based on DNA sequences and 16s rRNA sequence, C. burnetii was reclassified into the gamma subgroup of Proteobacteria, making it a close relative of species like Legionella pneumophila and Wolbachia persica.
Morphology and Microscopy of Coxiella burnetii
Coxiella burnetii is a small, pleomorphic, gram negative, obligate intracellular bacterium that typically appears as short rods or coccobacilli measuring approximately 0.2-0.4µm in width and 0.4-1.0µm in length. It possesses a typical germ negative cell envelope composed of an inner cytoplasmic membrane, a thin peptidoglycan layer and an outer membrane containing lipopolysaccharide (LPS).
The organism exhibits phase variation, where Phase I cells contain smooth LPS and are highly virulent, whereas Phase II cells possess truncated LPS and are less virulent, commonly arising after repeated laboratory passage. C. burnetii also demonstrates a biphasic developmental cycle consisting of a metabolically active large cell variant (LCV) and a dormant, environmentally resistant small cell variant (SCV) that allows survival under harsh environmental conditions. the bacterium is non-motile and non-spore-forming and lacks flagella and pili.
Microscopy
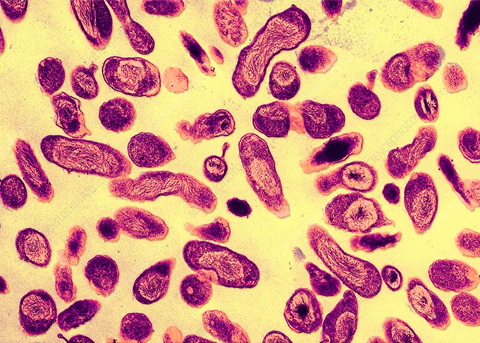
Coxiella burnetii is extremely small and difficult to visualize using routine light microscopy die to its intracellular nature and minute size. In infected host cells, the organism is typically observed within large membrane-bound vacuoles of the cytoplasm, often referred to as Coxiella-containing vacuoles. When stained with gram stain, the bacterium appears as poorly staining gram-negative coccobacilli and makes direct visualization challenging. Hence, special staining techniques such as Gimenez stain or modified Ziehl-Neelsen stain are commonly used to demonstrate the organism more clearly in infected tissues or cell cultures.
Under electron microscopy, Coxiella burnetii shows two morphological forms corresponding to its developmental cycle: the dense small cell variant and larger large cell variant within intracellular vacuoles. These microscopic characteristics reflect the bacteria’s adaptation to an intracellular lifestyle and its ability to survive in harsh intracellular environments.
https://www.cell.com/iscience/fulltext/S2589-0042%2823%2901287-7
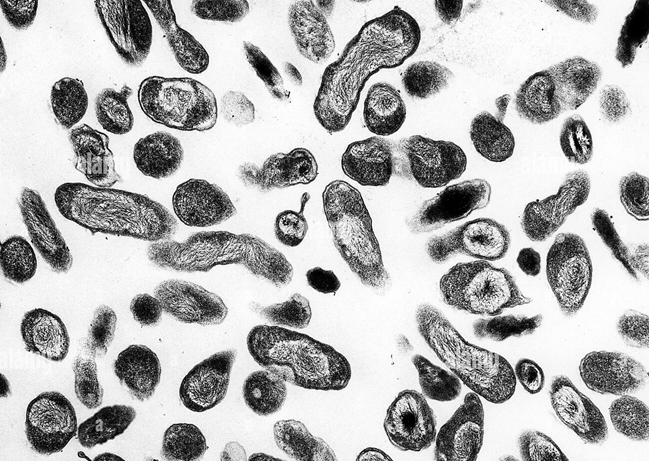
https://www.alamy.com/stock-photo-transmission-electron-micrograph-of-coxiella-burnetti-the-bacteria-30188590.html
Cultural and Growth Characteristics of Coxiella burnetii
Coxiella burnetii is a pleomorphic, gram negative, spore-forming coccobacillus. It is an obligate intracellular bacterium, meaning it cannot grow on conventional bacteriological culture media. The organism requires living host cells for replication and is typically cultivated in cell culture systems, embryonated chicken eggs or laboratory animals in a laboratory with full biosafety level 3 capability, due to its high infectivity and potential for aerosol transmission. In vitro, it can be propagated in several eukaryotic cell lines, including Vero and L929 cells where the bacteria multiply within large membrane-bound intracellular vacuoles.
Coxiella burnetii exhibits slow growth with a relatively long generation time compared with many other bacteria. The organism replicates within acidified phagolysosome like vacuoles of host cells, an unusual feature since most intracellular pathogens avoid such compartments. It demonstrates biphasic developmental cycle variant (LCV) responsible for replication and the dormant, environmentally resistant small cell variant (SCV) that facilitates survival under harsh environmental conditions. this ability to persist in resistant forms contributes to the bacterium’s environmental stability and high infectivity.
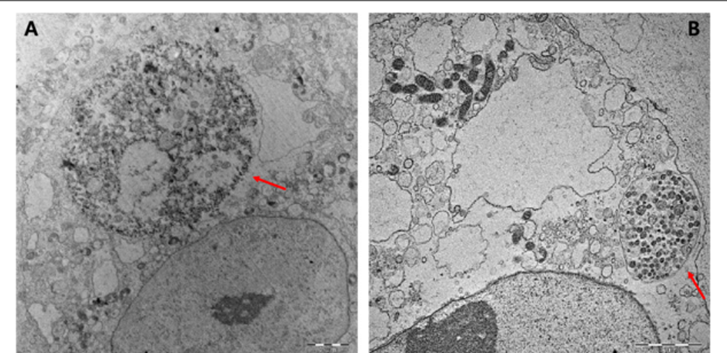
https://www.researchgate.net/publication/342254693_Isolation_of_Coxiella_burnetii_in_patients_with_nonspecific_febrile_illness_in_South_Korea
Epidemiology of Coxiella burnetii
C. burnetii is a bacterial agent responsible for Q fever and is a small negative bacterium with polymorphic properties. C. burnetii infection is a zoonotic disease that has a worldwide distribution and can manifest itself as a sporadic case or as outbreaks, except in a few regions such as New Zealand where the disease is rarely reported. The primary reservoirs of the bacterium are domestic ruminants, particularly sheep, goats and cattle although many other animals including wild mammals, birds and ticks can harbor the organism. Large numbers of bacteria are shed by infected animals in birth products, milk, urine and feces, leading to environmental contamination. Human infection most commonly occurs through inhalation of contaminated aerosols or dust particles originating from infected animals or their environments. The disease is therefore frequently associated with occupational exposure affecting individuals such as farmers, veterinarians, abattoir workers and laboratory personnel. Because C. burnetii can survive for long periods in the environment and is highly infectious even at very low doses, it can spread over long distances through windborne contaminated particles.
Pathogenesis and Virulence of Coxiella burnetii
Pathogenesis
C. burnetii is readily transmitted between hosts and environmental reservoirs, a characteristic that is partially attributed to the ability of the bacterium to survive the environment for long periods of time. The environmental stability of the organism seems to be linked to its ability to transition between distinct developmental stages. Small cell variants (SCVs) are metabolically inactive and resistant to numerous harsh environmental conditions. the switch from SCV to large cell variant (LCV) occurs after the invasion of host cells and during acidification of the phagosome, which triggers C. burnetii to become metabolically active.
The bacterium utilizes specialized secretion systems and virulence factors to manipulate host cell processes and maintain a favourable intracellular environment for replication. Following replication, infected macrophages can disseminate the organism through the bloodstream to various organs, particularly the liver, spleen, lungs and heart valves. In some individuals, persistent infection may lead to chronic Q fever, which is most commonly associated with endocarditis in patients with pre-existing valvular heart disease or immunocompromised conditions.
Virulence
C. burnetii possesses several virulence factors that enable it to survive and replicate within host cells. One of the most important virulence factors is its lipopolysaccharide (LPS), particularly the Phase I LPS, which is associated with virulent strains and helps the bacterium resist host immune responses. The organism also utilizes a Type IV secretion system that injects effector proteins into the host cells, allowing manipulation of host cellular processes and promoting intracellular survival.
Another key virulence trait is its ability to survive within acidic phagolysosome like vacuoles where most bacteria would normally be destroyed. This adaptation allows C. burnetii to exploit the host cell defense mechanisms for its own replication. The bacterium’s biphasic developmental cycle, involving the environmentally resistant SCV and the metabolically active LCV further enhances its ability to persist in both the environment and host tissues. SCV forms also show remarkable resistance to environmental stresses such as heat, desiccation and disinfectants, contributing to the organism’s long-term survival in contaminated dust and aerosols. These combined dust virulence mechanisms play a critical role in the organism’s infectivity, intracellular persistence and ability to cause both acute and chronic Q fever.
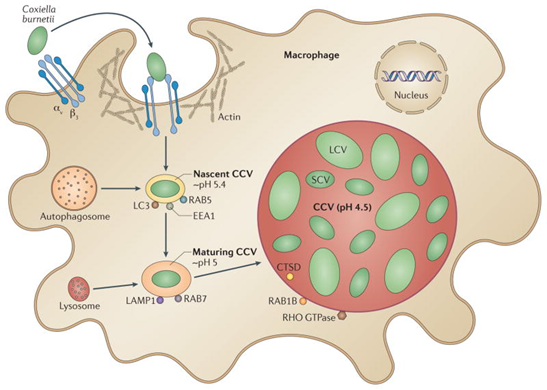
https://pmc.ncbi.nlm.nih.gov/articles/PMC4134018/
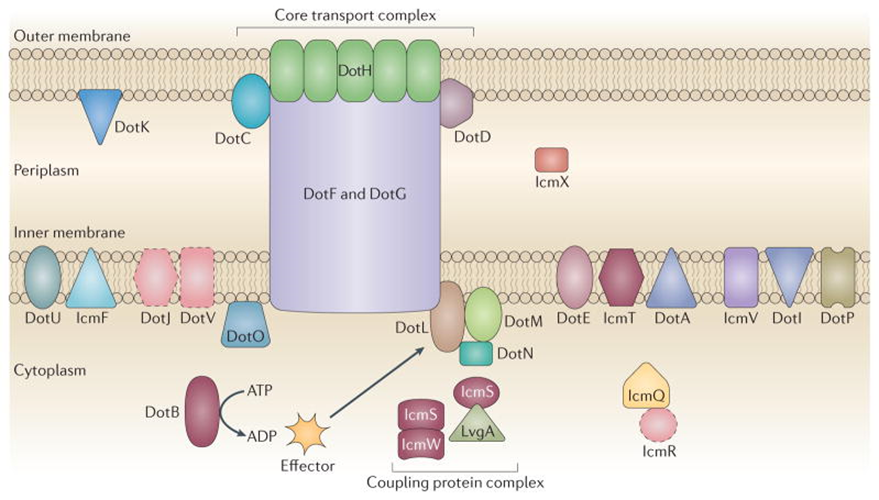
https://pmc.ncbi.nlm.nih.gov/articles/PMC4134018/
Clinical Manifestation of Coxiella burnetii
Q fever, caused by Coxiella burnetii, can cause both acute and chronic illness. People are commonly exposed from contact with infected animals or exposure to contaminated environments. Symptoms usually develop within 2-3 weeks of exposure, although as many as half of infected people are asymptomatic.
The following symptoms are commonly seen in patients with acute Q fever. However, it is important to note that the combination of signs and symptoms vary from person to person. Some patients may not have any symptoms following infection.
- Fever
- Fatigue
- Headache
- Malaise
- Myalgia
- Chills or sweats
- Cough
- Nausea
- Vomiting
- Diarrhoea
- Chest pain
In small proportion of patients, the infection progresses to chronic Q fever, particularly in individuals with pre-existing valvular heart disease, vascular grafts or immunocompromised conditions. the most common manifestation of chronic infection is endocarditis, which can lead to serious complications if untreated. Other chronic manifestations may include vascular infections, osteomyelitis, chronic hepatitis and interstitial lung disease. In addition, some patients experience neurological symptoms such as meningitis or encephalitis and a subset of individuals may develop post-Q fever fatigue syndrome, characterized by prolonged fatigue following acute infection.
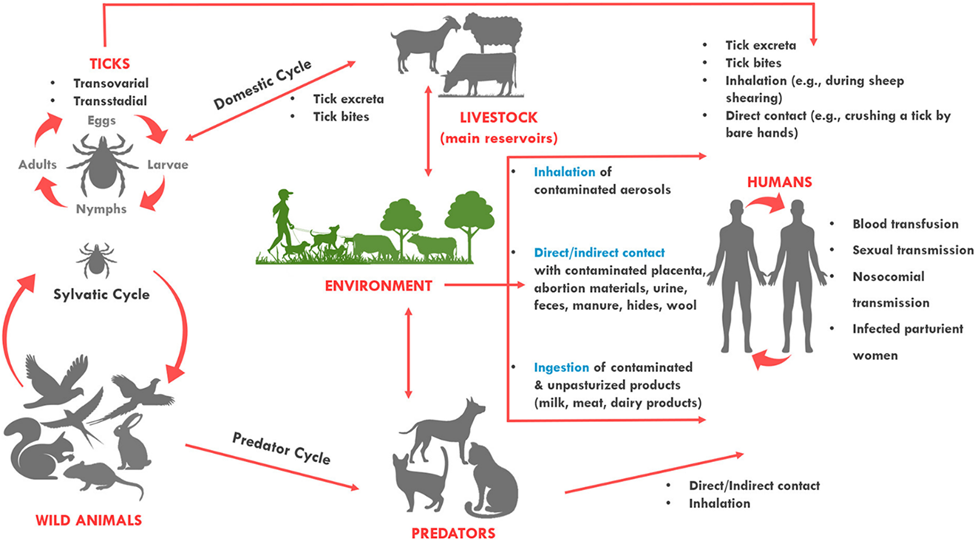
https://www.frontiersin.org/journals/veterinary-science/articles/10.3389/fvets.2022.1068129/full
Laboratory Diagnosis of Coxiella burnetii
Specimen: Blood, urine, feces, milk, yolk sac suspensions, contaminated laundry and clothing, naturally or experimentally infected animals, tissues of humans or other hosts
- Direct detection of Coxiella burnetii
Culture
the detection of C. burnetii in cell culture is performed on L929 fibroblasts or other cells such as Vero cells. For analysis, samples are extracted after filtration using 0.45 µm filters. Suitable for analysis are extracts from tissue samples and blood before the initiation of antibiotic treatment. Culturing C. burnetii must be performed under safety level 3 conditions to avoid the risk of laboratory contamination. Most recent studies show that C. burnetii can be cultured in acidified citrate cysteine (ACC) medium as Phase II Coxiella after an incubation period of around 6 days.
NAT- Nucleic Acid Amplification Test
Conventional PCR, nested PCR or Real-time PCR are used for the detection of C. burnetii nucleic acid. Primers are derived from the superoxide dismutase gene, plasmid or other gene segments. In addition, Coxiella DNA can be successfully detected in paraffin-embedded tissue. Real-time PCR with a sensitivity of 1 copy DNA has been developed. Coxiella can be detected in the blood by PCR 2 weeks after transmission. Reliable PCR detection methods for Coxiella have been used for genotyping.
- Serology
Antibodies
Besides IgM and IgG, IgA antibodies against C. burnetii can be detected. Antibodies become detectable 2-3 weeks after occurrence of clinical symptoms. They usually increase for some months and persist for years. High antibody titers against Phase I, which barely decrease over a period of 600 days, point to chronic course. IgM Phase II is still detectable 12 months after transmission of Coxiella in 62-82% of the blood samples.
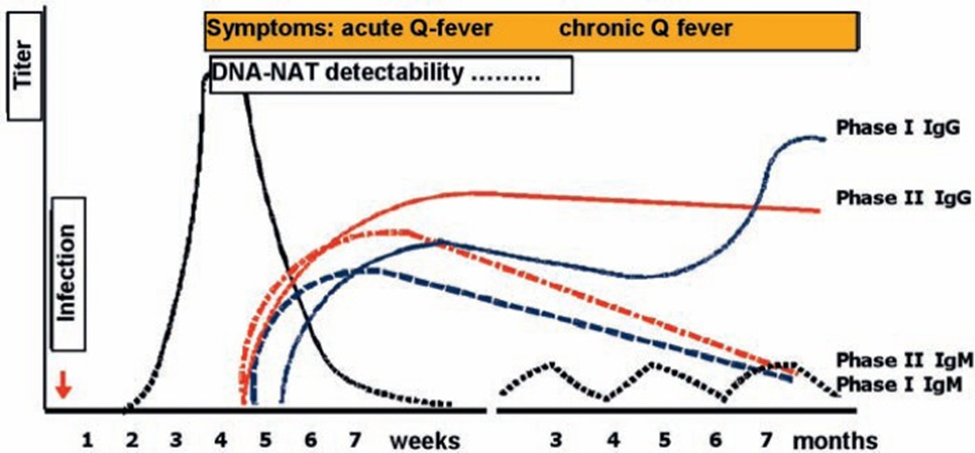
https://pmc.ncbi.nlm.nih.gov/articles/PMC3949614
Indirect Immunofluorescence Assay
The micro-immunofluorescence assay is considered the reference standard. The assay becomes positive around 1-2 weeks after appearance of the symptoms, additionally IgM antibodies can be detected using this method. Typically, tiers of ≥1:800 are considered positive for IgG for chronic Q fever and titers of ≥1:50 for IgM and ≥1:200 for IgG. As usual, seroconversion or titer levels that increase 4-fold are regarded as positive for the detection of an acute or reactivated infection.
ELISA, agglutination test and complement-fixation assay (CFA)
All 3 tests have been developed for routine diagnostics; CFA was most frequently used for early prevalence studies. CFA is regarded as positive if titers against Phase I are > 1:200 during chronic Q fever. Antibodies against Phase II antigens are considered as positive at ≥ 1:40 during acute Q fever. Due to high workload and the non-detectability of IgG2, IgG4 and IgA, as well as lacking standardization, CFA is nowadays replaced by ELISA or micro-immunofluorescence tests.
Western Blot
15 antigens are present on the strip of C. burnetii grown in Phase I, with a molecular weight of 20-160 kD. 7-10 different antigens may be stained with sera from the acute infection, especially the bands with 50, 80 and 160 kD; while 12-15 antigens may be stained with sera from the chronic phase.
Treatment of Coxiella burnetii
Acute Q fever
- First-line therapy
- Standard recommended treatment is Doxycycline 100 mg orally twice daily for ⁓14 days to shorten symptom duration and prevent complications.
- Should be started promptly on clinical suspicion without waiting for confirmatory tests because early therapy improves outcomes.
- Alternatives: Trimethoprim-sulfamethoxazole (TMP-SMX)- used in children <8 years or pregnant women when Doxycycline is contraindicated.
- Special considerations: Treatment generally not recommended for asymptomatic persons (unless high risk) because many cases self-resolve
Chronic Q fever
- Combination therapy
- For chronic Q fever manifestations (especially endocarditis or vascular infection), the backbone of treatment is Doxycycline + Hydroxychloroquine
- Hydroxychloroquine increases the pH of intracellular organelles, enhancing doxycycline’s bactericidal effect against C. burnetii
Duration of therapy: treatment is prolonged- often ≥18 months for endocarditis and may require monitoring of serologic titers and clinical response to determine exact duration.
Some regimens may include fluoroquinolones or rifampin for patient’s intolerant of hydroxychloroquine, but these are less well-studied.
Prevention of Coxiella burnetii
Preventing C. burnetii infection primarily involves minimizing exposure to the bacterium which Is commonly found in livestock and their birth products. Individuals working with animals such as farmers, veterinarians and abattoir workers should use protective clothing, gloves and masks to reduce inhalation of contaminated aerosols from birth products, urine or feces. Maintaining strict hygiene and disinfection protocols in farms and slaughterhouses is also essential to limit environmental contamination.
Vaccination is available in some countries, notably Australia, where Q-Vax vaccine is recommended for high-risk populations. Screening for past exposure is required prior to vaccination to prevent severe reactions. However, vaccination is not widely available globally.
Controlling the infection in animals is crucial for reducing human cases. Regular screening of livestock for C. burnetii, safe disposal of birth products and pasteurization of milk and dairy products significantly lower the risk of transmission.
Public awareness campaigns about avoiding contact with potentially contaminated animals or products, particularly during outbreaks, also play an important role in prevention.
References
- ASM. (2016). Coxiella burnetii. Clinical Microbiology Reviews, 12(4), 518–553. https://journals.asm.org/doi/10.1128/cmr.12.4.518
- ASM. (n.d.). Coxiella burnetii: Sentinel file. American Society for Microbiology. https://asm.org/asm/media/policy-and-advocacy/lrn/sentinel%20files/coxiella-burnetii.pdf
- Canada. (n.d.). Coxiella burnetii: Pathogen safety data sheet – infectious substances. Government of Canada. https://www.canada.ca/en/public-health/services/laboratory-biosafety-biosecurity/pathogen-safety-data-sheets-risk-assessment/coxiella-burnetii.html
- Centers for Disease Control and Prevention. (n.d.). Q fever: Clinical signs. https://www.cdc.gov/q-fever/hcp/clinical-signs/index.html#:~:text=symptoms%20following%20infection.-,Fever,Laboratory%20Testing%20for%20Q%20fever
- Frontiers in Veterinary Science. (2022). Coxiella burnetii in veterinary science. https://www.frontiersin.org/journals/veterinary-science/articles/10.3389/fvets.2022.1068129/full
- Microbe Notes. (n.d.). Coxiella burnetii: Characteristics, pathogenesis, diagnosis. https://microbenotes.com/coxiella-burnetii-characteristics-pathogenesis-diagnosis/
- NCBI Bookshelf. (n.d.). Coxiella burnetii. StatPearls. https://www.ncbi.nlm.nih.gov/books/NBK556095/
- NCBI Bookshelf. (n.d.). Coxiella burnetii infections. https://www.ncbi.nlm.nih.gov/books/NBK557893/
- PMC. (2014). Coxiella burnetii: Molecular microbiology insights. https://pmc.ncbi.nlm.nih.gov/articles/PMC3949614/
- PMC. (2014). Q fever epidemiology and clinical features. https://pmc.ncbi.nlm.nih.gov/articles/PMC4134018/
- PubMed. (2020). Coxiella burnetii infections: Clinical and laboratory analysis. https://pubmed.ncbi.nlm.nih.gov/32491816/
- ResearchGate. (2020). Isolation of Coxiella burnetii in patients with nonspecific febrile illness in South Korea. https://www.researchgate.net/publication/342254693_Isolation_of_Coxiella_burnetii_in_patients_with_nonspecific_febrile_illness_in_South_Korea
- Science Photo Library. (n.d.). Coxiella burnetii bacteria TEM. https://www.sciencephoto.com/media/612285/view/coxiella-burnetti-bacteria-tem
