- Coxsackie B viruses (CVB1–CVB6) are small, non-enveloped, positive-sense RNA enteroviruses that infect multiple organs (heart, pancreas, CNS, liver, pleura), causing both acute and chronic diseases.
- They are pantropic, with broad tissue tropism driven largely by usage of the Coxsackievirus and Adenovirus Receptor (CAR) and complement regulator decay-accelerating factor (DAF/CD55).
- Clinically, CBVs are major infectious causes of myocarditis, dilated cardiomyopathy, pleurodynia, pericarditis, pancreatitis, aseptic meningitis, encephalitis, neonatal sepsis-like disease, and are strongly implicated in type 1 diabetes pathogenesis.
- No licensed CBV-specific antiviral or vaccine exists, despite extensive evidence of disease burden and promising preclinical approaches.
Taxonomy and Classification
- Family: Picornaviridae
- Genus: Enterovirus
- Species: Enterovirus B (EV-B)
- Serotypes: Six classical CBV serotypes (CVB1–CVB6) within EV-B.
Key taxonomic points:
- EV-B species currently includes 63 serotypes (CVB1–6, CVA9, many echoviruses and other EV-B types).
- CBVs are classified serologically and by VP1 sequence; VP1 phylogeny underpins genotyping (e.g., CVB4 genotypes D/E, CVB5 genotypes A–D).
- CBV3 is the most frequently studied CV type in the literature, followed by CBV4 and CBV5, reflecting its central role in myocarditis and cardiomyopathy research.
Structure and Morphology
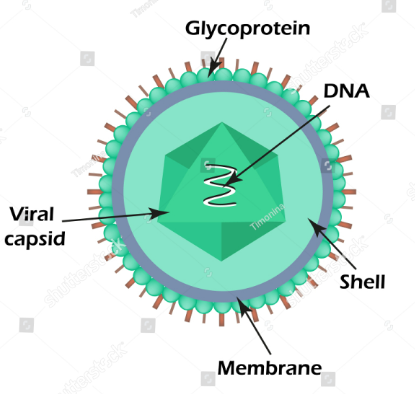
Figure: – Structure and Morphology of Coxsackie B viruses (Source: https://www.shutterstock.com/image-vector/structure-coxsackie-virus-enterovirus-infographics-vector-1367298365)
- Virion architecture
- Diameter ~30 nm, non-enveloped, icosahedral, T=pseudo-3 symmetry.
- Capsid built from 60 protomers, each containing VP1, VP2, VP3 (surface) and VP4 (internal).
- Capsid features
- Surface topography includes “canyon”, “puff”, and “knob” structures in CVB5, typical of EV-B; canyon often mediates receptor binding.
- A hydrophobic pocket beneath VP1 contains a lipid “pocket factor” whose release is associated with uncoating.
- VP4 lines the interior of the capsid and anchors to the RNA; cleavage of VP0 to VP2/VP4 marks mature virion formation.
- Structural states
- Full (F) particle: genome-containing, infectious virion.
- Altered (A) particle: expanded, receptor-primed uncoating intermediate.
- Empty (E) procapsid: RNA-free particle, assembly intermediate.
- High-resolution cryo-EM of CVB5 revealed conserved structural rearrangements from F → A → E, indicating a shared uncoating mechanism among EV-B.
Genome Organization and Proteins
- Genome
- ~7.4–7.5 kb positive-sense ssRNA.
- 5′ untranslated region (5′-UTR) with IRES and cloverleaf; 3′-UTR plus poly(A) tail.
- 5′ end covalently linked to VPg (3B), not capped.
- Polyprotein organization:
- Single ORF encodes large polyprotein → cleaved into:
- P1 (structural) → VP4, VP2, VP3, VP1.
- P2 (non-structural) → 2A, 2B, 2C.
- P3 (non-structural) → 3A, 3B (VPg), 3C, 3D.
- Single ORF encodes large polyprotein → cleaved into:
- Structural proteins
- VP1: outermost, most variable; major neutralizing antibody target and key determinant of antigenicity and receptor interaction.
- VP2/VP3: contribute to antigenic sites and capsid stability; additional neutralizing epitopes present.
- VP4: internal, myristoylated; important in RNA encapsidation and uncoating.
- Nonstructural proteins – functions:
- 2A protease: cleaves polyprotein and host eIF4G, shutting down host cap-dependent translation.
- 2B, 2C: membrane-active proteins driving formation of replication organelles, modulating Ca²⁺ homeostasis.
- 3A: anchors replication complex to intracellular membranes, interferes with secretory pathway.
- 3B (VPg): primer for RNA synthesis after uridylation.
- 3C protease: cleaves viral and numerous host proteins, modulating transcription, translation, innate immunity.
- 3Dpol: RNA-dependent RNA polymerase, highly error-prone, enabling quasispecies and recombination.
Replication Cycle
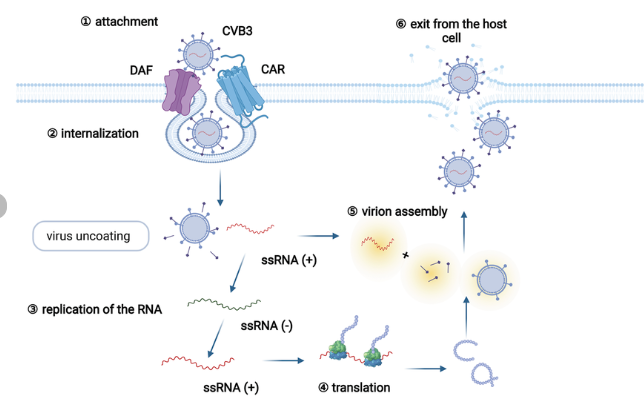
Figure: – Replication Cycle of Coxsackie B viruses (DOI: 10.3389/fphar.2022.843103)
Major stages
- Receptor binding & entry → uncoating → IRES-driven translation → polyprotein processing → RNA replication on modified membranes → assembly and maturation → cell lysis or vesicular release.
Attachment and Entry
- Primary receptors
- CAR (Coxsackievirus and Adenovirus Receptor): principal entry receptor for most CBVs; an immunoglobulin-like adhesion molecule localized at tight junctions and intercalated discs.
- DAF/CD55: complement regulator acting as an attachment receptor for many CBVs; concentrates virus on the cell surface and facilitates transfer to CAR.
- Entry process
- Virion binds DAF and/or CAR through VP1 canyon and adjacent loops, causing capsid conformational changes and pocket-factor expulsion.
- Internalization via clathrin-mediated endocytosis and possibly caveolar or macropinocytic routes, dependent on cell type.
- Acidification and/or receptor engagement promote transition to the A-particle, pre-uncoating state.
Uncoating and Genome Release
- F → A → E transition
- Full CVB5 particle expands to become A particle with:
- Externalization of VP4 and N-termini of VP1.
- Opening of channels at two-fold/three-fold axes.
- RNA genome exits into the cytosol; the A particle ultimately becomes an empty E particle.
- Full CVB5 particle expands to become A particle with:
- Cellular location
- Entire uncoating and subsequent replication remain cytoplasmic; no nuclear phase.
Translation and Polyprotein Processing
- Immediate translation
- Positive-sense RNA functions as mRNA; 5′ IRES recruits ribosomes in a cap-independent manner.
- Translation initiates a single large polyprotein that is co- and post-translationally cleaved by viral proteases.
- Host shutoff
- 2Apro and 3Cpro cleave host:
- eIF4G, PABP, and other translation factors → global inhibition of cellular protein synthesis.
- Components of innate immunity (e.g., MAVS, TRIF) → suppression of IFN production.
- 2Apro and 3Cpro cleave host:
Formation of Replication Organelles & RNA Replication
- Membrane remodeling
- Nonstructural proteins recruit and remodel ER/Golgi membranes into double-membrane vesicles that serve as replication organelles.
- Initiation
- VPg is uridylylated by 3Dpol to form VPg-pUpU, which primes negative-strand synthesis.
- Replication
- Negative-strand RNA is synthesized on the genomic positive strand to form dsRNA intermediates.
- Multiple positive strands are produced from each negative template, massively amplifying genome copies.
- Genetic variability
- High mutation rate and frequent recombination with other EV-B types (e.g., in CVB4 and CVB5 P2/P3 regions) generate diverse recombinant lineages.
Assembly, Maturation and Release
- Assembly
- P1 products assemble into protomers → pentamers → RNA-free procapsids, which package viral RNA.
- Maturation cleavage
- VP0 cleaves into VP2 + VP4 in RNA-containing particles, stabilizing virions and enabling infectivity.
- Release
- Cell lysis following extensive cytopathic effect is the predominant mechanism; virions are released into extracellular space.
- CBVs can also exit non-lytically in extracellular microvesicles, providing quasi-enveloped particles that evade neutralizing antibodies and disseminate systemically.
Pathogenesis and Host Immune Response
Determinants of Tropism and Disease
- Pan tropism
- CBVs infect heart, pancreas, CNS, liver, skeletal muscle, pleura, and neonatal organs, explaining their broad clinical spectrum.
- Receptor expression
- CAR is abundantly expressed in cardiomyocytes, pancreatic β-cells, intestinal epithelium, CNS endothelium and many tumors, aligning with cardiac, pancreatic and neurotropism.
Direct Viral Cytotoxicity
- Acute injury
- Productive lytic replication causes myocyte necrosis, β-cell death, neuronal death, and hepatocellular damage.
- Viral proteases and nonstructural proteins disrupt cytoskeleton, mitochondrial integrity, and cell–cell junctions, promoting cell death.
Innate Immune Responses
- Pattern recognition
- Viral RNA recognized by MDA5, RIG-I, TLR3, triggering type I IFN and inflammatory cytokines.
- Immune evasion
- 2Apro and 3Cpro cleave MAVS, TRIF, and other signaling molecules, attenuating IFN induction.
- Infection of immune cells and lymphoid tissue contributes to transient immunosuppression.
Adaptive Immunity and Immunopathology
- Humoral immunity
- Neutralizing IgG and IgA against capsid proteins (especially VP1) are type-specific and crucial for viral clearance and long-term protection.
- Cell-mediated immunity
- CD8⁺ T cells recognize multiple capsids and non-capsid epitopes; immune pressure is exerted across the genome, not restricted to P1.
- Ex vivo stimulation of PBMCs with CBV3 peptides shows broad IFN-γ responses to multiple peptide pools, indicating diversified effector memory targets.
- Autoimmunity and chronic disease
- In myocarditis and type 1 diabetes, CBVs are thought to trigger:
- Molecular mimicry and autoimmune responses against cardiac or β-cell antigens.
- Persistent low-level infection with ongoing immune-mediated tissue damage.
- In myocarditis and type 1 diabetes, CBVs are thought to trigger:
Epidemiology and Transmission
- Global distribution
- CBVs are worldwide and highly prevalent, especially in children; CBV3, CBV4, and CBV5 dominate the literature and sequence databases.
- Transmission routes
- Fecal–oral and respiratory spread; virus replicates in gut and oropharynx, then disseminates hematogenously.
- Vertical and perinatal transmission can cause fulminant neonatal disease.
- Molecular epidemiology
- CBV4:
- Frequent recombination with multiple EV-B serotypes; 12 recombinant patterns (A–L) identified across global genomes.
- VP1 genotypes D (outside China) and E (dominant in China) with rapid evolution (>6.5×10⁻³ substitutions/site/year).
- CBV5:
- Four VP1 genotypes (A–D) globally; in China, genotypes B and D co-circulate with at least two major lineages and frequent P2/P3 recombination.
- CBV4:
Clinical Manifestations
General spectrum
- Mild, self-limited disease
- Fever, upper respiratory tract infections (URTI), gastroenteritis, nonspecific febrile illness.
- Severe acute diseases
- Myocarditis, dilated cardiomyopathy, pleurodynia, pericarditis, pancreatitis, hepatitis, aseptic meningitis, encephalitis, myelitis, acute flaccid paralysis, systemic neonatal infection.
- Chronic and immune-mediated conditions
- Chronic myocarditis / cardiomyopathy and type 1 diabetes mellitus strongly associated with CBV infections.
Cardiac Disease
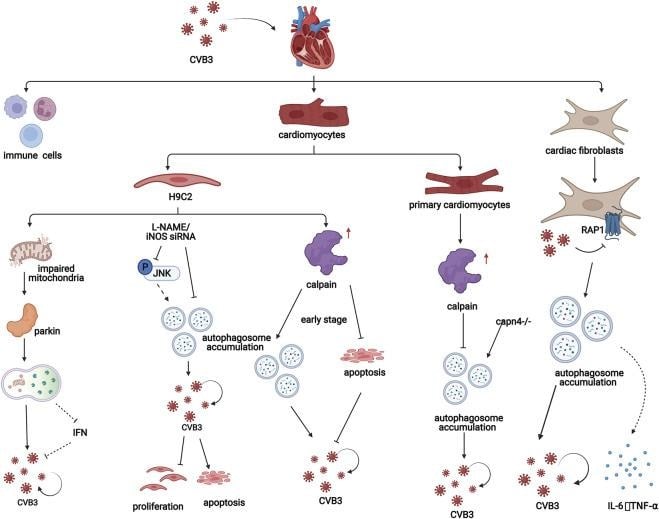
Figure: In viral myocarditis, CVB3 influences the process of autophagy in both cardiomyocytes and cardiac fibroblasts (https://doi.org/10.3389/fphar.2022.843103)
- Acute myocarditis & pericarditis
- CBV3 is prototypic; CBV1, CBV2, CBV4, CBV5, CBV6 also implicated.
- Clinical features:
- Chest pain, dyspnea, arrhythmias, heart failure; ECG changes and elevated troponin.
- Animal models:
- CVB3-Nancy in mice induces acute myocarditis and dilated cardiomyopathy; chronic models show persistent viral RNA and autoimmune responses.
- Dilated cardiomyopathy (DCM)
- CBV3 and CBV4 can establish persistent infection in myocardium, with low-level replication and immune-mediated damage leading to DCM.
Pancreatic Disease and Type 1 Diabetes
- Acute pancreatitis
- CBV1–CBV5 cause acute pancreatitis in humans and severe/chronic pancreatitis in mouse models (e.g., CBV4-V).
- Type 1 diabetes mellitus (T1DM)
- Epidemiologic, serologic, and animal data show strong association of CBV (especially CBV4) with T1DM.
- Viral replication in β-cells (using CAR) induces cell death and may trigger autoimmune β-cell destruction.
- CBV-targeted vaccines are under development as T1DM preventive strategies.
Central Nervous System (CNS) Disease
- Acute CNS infection
- CBVs cause aseptic meningitis, encephalitis, meningoencephalitis, myelitis, and acute flaccid paralysis, mainly in newborns and children.
- CBV5 is a frequent cause of aseptic meningitis and encephalitis; Brazilian surveillance (2005–2018) identified CVB5 in CSF and stool of CNS-disease patients with distinct genogroups and capsid substitutions affecting tropism and antigenicity.
- Neurodegenerative/psychiatric associations
- Experimental and epidemiological studies suggest possible CBV involvement in amyotrophic lateral sclerosis and some psychiatric disorders, though mechanisms and causality remain unclear.
Neonatal and Perinatal Disease
- Systemic neonatal infection
- Infants <3 months, especially neonates infected intrapartum, can develop:
- Fulminant myocarditis, hepatitis, meningoencephalitis, sepsis-like syndrome, DIC, multi-organ failure, and high mortality.
- Infants <3 months, especially neonates infected intrapartum, can develop:
Other Manifestations
- Pleurodynia (Bornholm disease)
- Sharp, pleuritic chest or abdominal pain with fever; classic CBV-associated syndrome.
- HFMD and mucocutaneous disease
- CBV can occasionally cause HFMD, though Coxsackie A and EV-A71 predominate.
Laboratory Diagnosis
- Direct detection
- RT-PCR:
- Pan-enterovirus PCR targeting 5′-UTR followed by VP1 sequencing for typing; applied directly to CSF, blood, stool, throat swabs.
- Quantitative RT-PCR used for viral load in myocardium, pancreas, or CSF in research settings.
- RT-PCR:
- Virus isolation
- Culture from clinical specimens in RD, HEp-2, Vero, or human fibroblast/heart cell lines for phenotypic and pathogenesis studies.
- Serology
- Neutralization tests and ELISA detect type-specific CBV antibodies, useful in epidemiology and retrospective diagnosis, particularly when RNA is no longer detectable.
- Histopathology and molecular pathology
- In biopsy/autopsy tissues (e.g., heart, pancreas):
- In situ hybridization or RT-PCR for viral RNA.
- Immunohistochemistry for viral proteins.
- Correlation with inflammatory infiltrates supports etiologic role in myocarditis or T1DM.
- In biopsy/autopsy tissues (e.g., heart, pancreas):
Treatment and Antiviral Therapy
Current Clinical Management
- No licensed CBV-specific antivirals or vaccines.
- Management is largely supportive:
- Heart failure therapy for myocarditis/DCM.
- ICU care for neonatal sepsis-like disease or severe CNS infection.
Antiviral Strategies Under Investigation
- Direct-acting antivirals
- Capsid binders (pocket-factor mimetics) and 3Dpol inhibitors have shown in vitro and animal efficacy against CBVs.
- Gemcitabine + ribavirin combination exhibited synergistic antiviral activity against enteroviruses (including CBV3) in vitro.
- Host-targeted agents
- Modulators of autophagy, PI3K, MAPK, and other pathways exploited by CBVs can reduce viral replication.
- Transcriptomic analyses in CBV3-infected brain endothelial cells identified MEK, PDGFR and VEGF inhibitors (e.g., pelitinib, neratinib) as repurposing candidates.
Vaccines
- Experimental vaccines
- RNA vaccine against CBV3 protected mice from lethal challenge, reducing viral load and myocarditis.
- Virus-like particle (VLP) vaccines:
- Modified CVB VLP induced high levels of neutralizing antibodies and strong Th1-biased T-cell responses in mice; adjuvanted VLPs elicited robust IFN-γ and IL-2 secretion.
- Multi-epitope vaccines:
- Immunoinformatic-designed multivalent epitope constructs targeting VP1 and nonstructural proteins predicted to be antigenic, non-allergenic, and stable, with favorable docking to MHC and TLRs.
- T1DM-oriented vaccines
- Strategies targeting CBV, especially CBV1/CBV4, are being pursued as preventive vaccines against type 1 diabetes, with promising preclinical and early clinical developments.
Oncolytic Virotherapy
- CBV3 and other CBVs as oncolytic agents
- CBV3 selectively replicates in and lyses CAR- and heparan sulfate–expressing tumors, inducing direct oncolysis and robust anti-tumor immune responses in multiple mouse cancer models.
- Genetic engineering (e.g., insertion of tumor-specific miRNA target sites) can attenuate cardiotoxicity while preserving oncolytic efficacy.
Prevention and Control
- General measures
- Hand hygiene, disinfection of contaminated surfaces, and avoidance of close contact during outbreaks.
- Infection control in neonatal units and maternity wards to prevent perinatal CBV transmission.
- Surveillance
- Molecular typing of enterovirus isolates (including CBVs) from HFMD, CNS disease, and myocarditis improves understanding of circulating genotypes, recombination patterns, and geographic spread.
- Vaccine prospects
- Multivalent CBV vaccines (e.g., VLP or epitope-based) are under preclinical/early clinical development for myocarditis/DCM and T1DM prevention, and may also reduce neonatal and CNS disease burden.
Conclusion
Coxsackie B viruses are prototypical pantropic enteroviruses with a compact yet versatile genome and a highly structured capsid that together support efficient replication, broad tissue tropism, and significant pathogenic potential. Through use of CAR and DAF, CBVs infect heart, pancreas, CNS and other organs, producing diseases that range from mild febrile illness to life-threatening myocarditis, neonatal sepsis, and chronic autoimmune conditions such as type 1 diabetes. Their replication cycle relies on IRES-mediated translation, protease-driven host shutoff, formation of membrane-bound replication organelles, and high-error RNA replication that fuels genetic diversity and recombination. Although current management is largely supportive, rapid advances in antiviral discovery, structure-guided vaccine design, immunoinformatic, and oncolytic virotherapy position CBVs as both important human pathogens and promising therapeutic tools. Effective vaccines and targeted antivirals against CBV could have major implications not only for acute infections but also for prevention of chronic sequelae like cardiomyopathy and autoimmune diabetes.
References
- Gad, B., & Wieczorek, M. (2023). Coxsackie B – pantropic viruses. Postępy Mikrobiologii – Advancements of Microbiology, 62(1), 13–26. https://doi.org/10.2478/am-2023-0002
- Alhazmi, A., Nekoua, M. P., Mercier, A., Vergez, I., Sane, F., Alidjinou, E. K., & Hober, D. (2022). Combating coxsackievirus B infections. Reviews in Medical Virology, 33(1), e2406. https://doi.org/10.1002/rmv.2406
- Weng, S., Zhu, R., Wu, Y., Xia, N., Xu, L., & Cheng, T. (2024). Research progress and application prospects of animal models of group B Coxsackievirus infections. Emerging Microbes & Infections, 14(1), 2441391. https://doi.org/10.1080/22221751.2024.2441391
- Liu, H., & Luo, H. (2021). Development of Group B Coxsackievirus as an oncolytic virus: opportunities and challenges. Viruses, 13(6), 1082. https://doi.org/10.3390/v13061082
- Xu, D. H., Zhang, M., Chen, J. W., Feng, C. Z., Liu, Y. H., Chu, Z. Y., Liu, W. J., Li, L., & Hui, S., MA. (2024). Molecular Characterization of Coxsackievirus B1 Strains Isolated from Patients with Hand Foot and Mouth Disease in Yunnan, Southwest China. PubMed, 37(5), 543–548. https://doi.org/10.3967/bes2024.061
- Jmii, H., Fisson, S., Aouni, M., & Jaidane, H. (2020). Type B coxsackieviruses and central nervous system disorders: critical review of reported associations. Reviews in Medical Virology, 31(4), e2191. https://doi.org/10.1002/rmv.2191
- Xiao, J., Wang, J., Lu, H., Song, Y., Sun, D., Han, Z., Li, J., Yang, Q., Yan, D., Zhu, S., Pei, Y., Wang, X., Xu, W., & Zhang, Y. (2023). Genomic epidemiology and transmission dynamics of global Coxsackievirus B4. Viruses, 15(2), 569. https://doi.org/10.3390/v15020569
- Kundu, R., Knight, R., Dunga, M., & Peakman, M. (2018). In silico and ex vivo approaches indicate immune pressure on capsid and non-capsid regions of coxsackie B viruses in the human system. PLoS ONE, 13(6), e0199323. https://doi.org/10.1371/journal.pone.0199323
- Dutkiewicz, M., Swiatkowska, A., Ojdowska, A., Smólska, B., Dymarek-Babś, T., Jasińska, A., & Ciesiołka, J. (2014). REVIEW PAPER<br>Molecular mechanisms of genome expressionof coxsackievirus B3 that belongs to enteroviruses. BioTechnologia, 93(4), 414–423. https://doi.org/10.5114/bta.2012.46595
- Garmaroudi, F. S., Marchant, D., Hendry, R., Luo, H., Yang, D., Ye, X., Shi, J., & McManus, B. M. (2015). Coxsackievirus B3 replication and pathogenesis. Future Microbiology, 10(4), 629–653. https://doi.org/10.2217/fmb.15.5
- Machado, R. S., Gomes-Neto, F., Aguiar-Oliveira, M. L., Burlandy, F. M., Tavares, F. N., Da Silva, E. E., & Sousa, I. P. (2022). Analysis of Coxsackievirus B5 Infections in the Central Nervous System in Brazil: Insights into Molecular Epidemiology and Genetic Diversity. Viruses, 14(5), 899. https://doi.org/10.3390/v14050899
- Huang, S., Zhang, C., Li, J., Dai, Z., Huang, J., Deng, F., Wang, X., Yue, X., Hu, X., Li, Y., Deng, Y., Wang, Y., Wang, Y., Zhao, W., Zhong, Z., Wang, Y., & Wang, Y. (2022). Designing a multi-epitope vaccine against coxsackievirus B based on immunoinformatics approaches. Frontiers in Immunology, 13, 933594. https://doi.org/10.3389/fimmu.2022.933594
- He, Y., Wei, H., Wei, L., Fan, H., Yan, D., Zhao, H., Zhu, S., Ji, T., Xiao, J., Lu, H., Wang, W., Guo, Q., Yang, Q., Xing, W., & Zhang, Y. (2022). Molecular epidemiology reveals the Co-Circulation of two genotypes of Coxsackievirus B5 in China. Viruses, 14(12), 2693. https://doi.org/10.3390/v14122693
- Soppela, S., González-Rodríguez, M., Stone, V. M., Mustonen, I., Jouppila, N. V. V., Lampinen, V., Haikarainen, T., Flodström-Tullberg, M., Junttila, I. S., & Hankaniemi, M. M. (2025). Coxsackie B1 virus-like particle vaccine modified to exclude a highly conserved immunoreactive region from the capsid induces potent neutralizing antibodies and protects against infection in mice. Journal of Biomedical Science, 32(1), 86. https://doi.org/10.1186/s12929-025-01183-1
- Yang, P., Shi, D., Fu, J., Zhang, L., Chen, R., Zheng, B., Wang, X., Xu, S., Zhu, L., & Wang, K. (2022). Atomic structures of Coxsackievirus B5 provide key information on viral evolution and survival. Journal of Virology, 96(9), e0010522. https://doi.org/10.1128/jvi.00105-22
- Machado, R. S., Tavares, F. N., & Sousa, I. P. (2024). Global landscape of coxsackieviruses in human health. Virus Research, 344, 199367. https://doi.org/10.1016/j.virusres.2024.199367
- Wood, J. F., Vergis, J. M., Imami, A. S., Ryan, W. G., Sin, J. J., Kim, B. J., Schiefer, I. T., & McCullumsmith, R. E. (2025). Identification of drug repurposing candidates for Coxsackievirus B3 infection in IPSC-Derived brain-like endothelial cells. International Journal of Molecular Sciences, 26(15), 7041. https://doi.org/10.3390/ijms26157041
- Geisler, A., Hazini, A., Heimann, L., Kurreck, J., & Fechner, H. (2021). Coxsackievirus B3—Its potential as an oncolytic virus. Viruses, 13(5), 718. https://doi.org/10.3390/v13050718
- Tracy, S., Chapman, N. M., & Beck, M. A. (1991). Molecular biology and pathogenesis of coxsackie B viruses. Reviews in Medical Virology, 1(3), 145–154. https://doi.org/10.1002/rmv.1980010304
- Yu, K., Zhou, L., Wang, Y., Yu, C., Wang, Z., Liu, H., Wei, H., Han, L., Cheng, J., Wang, F., Wang, D. W., & Zhao, C. (2022). Mechanisms and therapeutic Strategies of Viral myocarditis targeting Autophagy. Frontiers in Pharmacology, 13, 843103. https://doi.org/10.3389/fphar.2022.843103
