Mycoplasma pneumoniae is a bacterium belonging to the class Mollicutes, that can infect humans. It usually causes upper respiratory tract infections but can also cause pneumonia. Many extrapulmonary infections have been attributed to M. pneumoniae infections.
The bacteria typically cause self-limiting respiratory infections and is commonly referred to as “walking pneumonia”. M. pneumoniae is a frequent cause of respiratory tract infections on both children and adults, that can range in severity from mild to life-threatening. It is sometimes associated with extrapulmonary manifestations involving multiple organ systems and this organism has been linked to development and/or exacerbations of asthma.
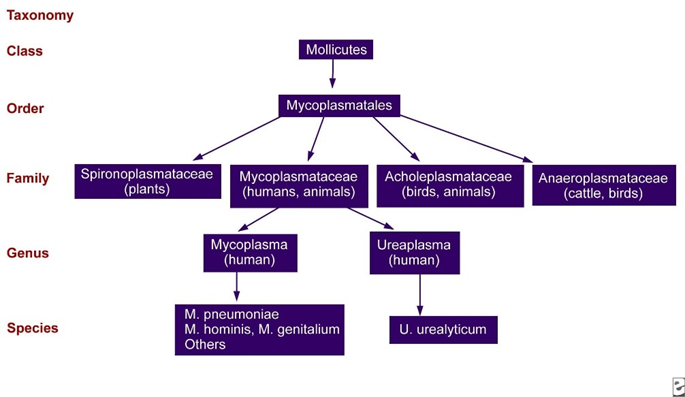
Classification of Mycoplasma pneumoniae
Domain: Bacteria
Phylum: Mycoplasmatota
Class: Mollicutes
Order: Mycoplasmatales
Family: Mycoplasmataceae
Genus: Mycoplasma
Species: Mycoplasma pneumoniae
Members of class Mollicutes are distinguished by their lack of peptidoglycan, small genome size, sterol-containing cell membranes and pleomorphic morphology.
https://emedicine.medscape.com/article/1941994-overview#a1
Morphology and Microscopy of Mycoplasma pneumoniae
Morphology
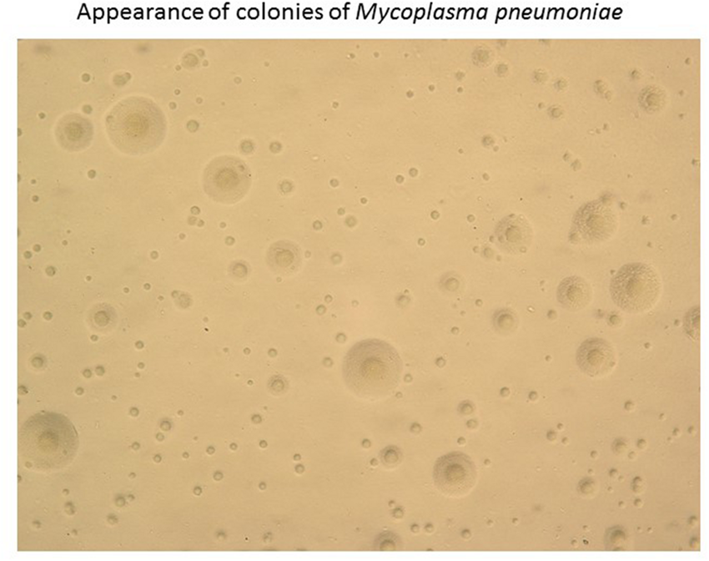
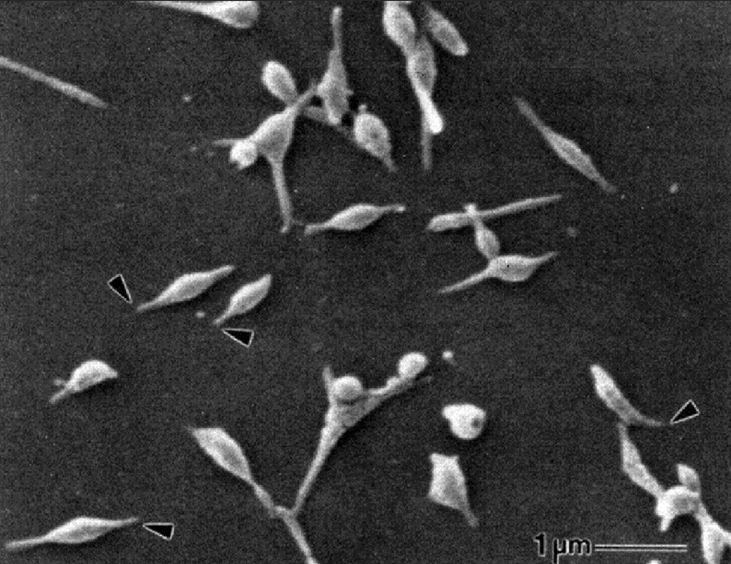
Mycoplasma pneumoniae is a spindle shaped pleomorphic bacterium that measures around 1-2 µm long by 0.1-0.2 µm wide. They exhibit gliding motility on liquid-covered surfaces. The tip of the structure always leads, indicating its importance in attachment. M. pneumoniae lacks a cell wall and has a three-layered membrane containing sterols that provide structural support. The cell membrane is supported by a protein network which creates a cytoskeleton. M. pneumoniae colonies are up to 100 µm in diameter, primarily colonizing the respiratory tract. One of the most useful distinguishing features of mycoplasmas is their peculiar fried-egg colony shape, consisting of a central zone of growth embedded in the agar (specialized media like Eaton’s agar) and a peripheral one on the agar surface, although the growth is slow.
Microscopy
M. pneumoniae can adhere to glass and plastic surfaces and the morphology of them growing on these surfaces has been studied.
- Light Microscopy: M. pneumoniae cannot be visualized by gram staining due to the absence of a cell wall and its extremely small size. It does not retain conventional bacterial stains reliably.
- Electron Microscopy: Transmission electron microscopy reveals the characteristic flask-shaped structure and the terminal attachment organelle. The trilaminar membrane structure can also be observed.
The organisms’ minimal genome and lack of rigid structural components contribute to its small size, pleomorphism and fastidious growth requirements.
https://www.researchgate.net/figure/Appearance-of-colonies-of-Mycoplasma-pneumoniae-Colonies-of-M-pneumoniae-on-an-agar_fig2_299344460
https://www.researchgate.net/figure/Scanning-electron-micrograph-of-M-pneumoniae-cell-reprinted-from-reference-9_fig1_286932576
Cultural and Growth Characteristics of Mycoplasma pneumoniae
Mycoplasma pneumoniae is a fastidious, slow-growing organism that requires specialized media enriched with sterols (such as cholesterols), which are essential for membrane synthesis due to its lack of cell walls. A routine mycoplasma medium consists of heart infusion, peptone, yeast extracts, salt, glucose or arginine and horse serum/ to prevent the overgrowth of the fast-growing bacteria that usually accompany mycoplasmas in clinical materials, penicillin, thallium acetate or both are added as selective agents. When M. pneumoniae grows in the medium, it produces acid, causing the color of the medium to change from purple to yellow. Commonly used media include Eaton’s agar or broth supplemented with serum and yeast extract.
Growth is aerobic to facultatively anaerobic and occurs optimally at 35-37°C with increased humidity which is consistent with its adaptation to the human respiratory tract. Colonies typically occur after 1-2 weeks of incubation, telling of its slow replication rate.
On solid media, colonies display the characteristic “fried egg” morphology consisting of a dense central core embedded in the agar and a lighter peripheral zone spreading on the surface. This appearance results from the organism’s ability to growth within and on the agar surface. In liquid media, growth produces slight turbidity and may cause a pH change due to glucose fermentation, which can be detected using phenol red indicator. However, due to the prolonged incubation time (4 days to over 23 days) and low sensitivity, routine culture is rarely performed in clinical practices. Instead, molecular methods such as PCR are preferred for diagnosis.
Epidemiology of Mycoplasma pneumoniae
M. pneumoniae is a globally distributed respiratory pathogen and a significant cause of community-acquired pneumonia (CAP), particularly among school-aged children, adolescents and young adults. It is transmitted from one person to another via respiratory droplets (when they cough or sneeze) during close and prolonged contact. It may also be spread by direct contact with the nose or throat discharges of an infected person or indirectly through contact with articles freshly soiled by secretions of infected people. It has an incubation period that ranges between 2 to 3 weeks. Like most respiratory pathogens, infection usually occurs during the winter months but can happen year-round. Outbreaks of mycoplasma infection occur in military recruits, hospitals, nursing homes and other crowded or semi-closed environments where the close contact for prolonged time is a frequent occurrence.
Although respiratory disease is the primary manifestation, M. pneumoniae infections may also lead to extrapulmonary complications involving the skin, nervous system, cardiovascular system and hematologic system, though these occur less frequently. Only 5-10% of people infected with mycoplasma develop pneumonia. It causes upper and lower respiratory tract infections.
Pathogenesis and Virulence Factors of Mycoplasma pneumoniae
The organism responsible for mycoplasmal pneumonia, M. pneumoniae is a pleomorphic organism that unlike bacteria, lacks a cell wall and unlike viruses, does not need a host cell for replication. They are primarily mucosal pathogens, living a parasitic existence in close association with epithelial cells of their host, usually in the respiratory or urogenital tracts. Adherence is firm enough to prevent the elimination of the parasites by mucous secretions or urine.
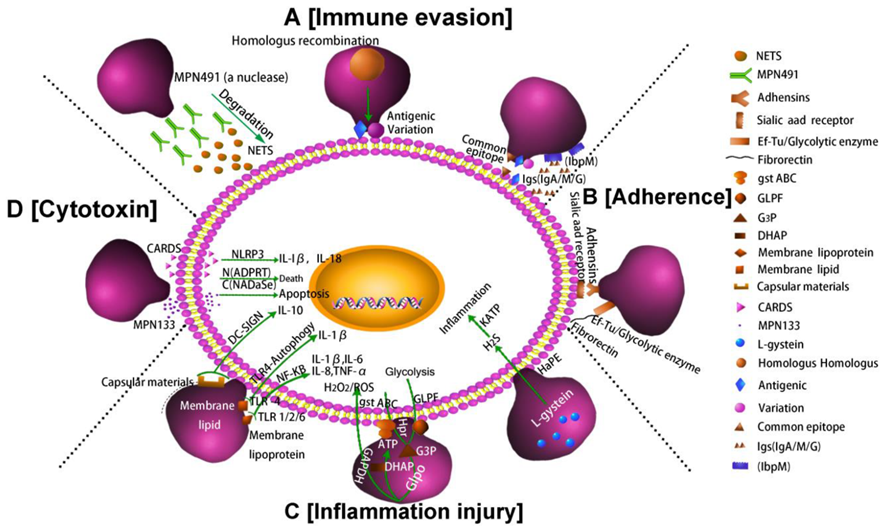
The pathogenicity of M. pneumoniae is primarily associated with its ability to attach to respiratory epithelial cells, persist within host tissues and induce cellular damage and inflammation. The main mechanisms involved in its pathogenesis include cytadherence, intracellular localization, cytotoxicity and inflammatory response.
- Cytadherence
Attachment of Mycoplasma pneumoniae to the respiratory epithelium is a crucial initial step in the development of disease. Close adherence allows the organism to resist removal by the host’s mucociliary clearance system and enables it to exert localized cytotoxic effects on epithelial cells. To achieve this, M. pneumoniae possesses a specialized terminal attachment organelle visible under electron microscope, consisting of a tip structure with a dense central filament surrounded by membrane extensions. This structure contains a network of adhesins and accessory proteins that coordinate attachment to the host cell receptors such as soaloglycoconjugates, sulfated glycolipids and fibronectin. The P1 adhesin (surface protein concentrated at the attachment tip) plays the primary role on mediating adhesion to host cells. Loss or mutation of this protein markedly reduces adherence and virulence. However, effective cytadherence requires cooperative action of additional proteins, including P30, HMW1-HMW5, P90 and P65, which contribute to the formation and function of attachment organelle and facilitate stable colonization of the respiratory epithelium.
- Intracellular Localization
Although M. pneumoniae is primarily an extracellular mucosal pathogen that resides on epithelial surfaces, studies have shown that some mycoplasmas can fuse with and enter non-phagocytic host cells, a capability likely facilitated by their lack of cell wall. experimental evidence indicates that M. pneumoniae can survive, synthesize DNA, and replicate within artificial cell culture systems for extended periods. Such intracellular localization may enable the organism to evade immune defenses, persist in latent or chronic infections, cross mucosal barriers and reduce the effectiveness of certain antimicrobial therapies. Fusion between the mycoplasmal and host cell membranes may also release hydrolytic enzymes and insert mycoplasmal membrane components into host membranes, potentially altering receptor recognition and cytokine responses. However, the extent to which M. pneumoniae invades and replicates intracellularly in vivo and the clinical significance of these processes remain uncertain.
- Cytotoxicity and Inflammation
Following the attachment of M. pneumoniae to respiratory epithelial cells, cellular injury occurs primarily through localized cytotoxic effects and host inflammatory responses, even without intracellular invasion. The organism does not produce classical exotoxins. Instead, it generates reactive oxygen species, particularly hydrogen peroxide and superoxide radicals, which include oxidative stress and damage host cell components such as lipids, protein and membranes. Because M. pneumoniae lacks protective enzymes like catalase and superoxide dismutase, these reactive molecules accumulate and contribute to hemolysis and epithelial injury. Additional mechanisms, such as the acquisition of host lactoferrin and iron complexes, may further enhance oxidative damage through the production of highly reactive hydroxyl radicals. Infection leads to structural and functional damage to respiratory cilia, reduced cellular metabolism and eventual exfoliation of epithelial cells, resulting in impaired mucociliary clearance and the characteristic persistent cough associated with infection. Once the organism reaches the lower respiratory tract, immune responses are triggered, including activation of macrophages, recruitment of neutrophils and lymphocytes and release of pro-inflammatory cytokines such as TNF-α, IFN-γ, IL-1β, IL-6 and IL-8. While these immune reactions help control infection, excessive cytokine production and lymphocyte activation can contribute to pulmonary inflammation and tissue injury, indicating that both direct microbial effects and immune-mediated mechanism play roles in pathogenesis of M. pneumoniae diseases.
https://www.mdpi.com/2076-0817/10/2/119
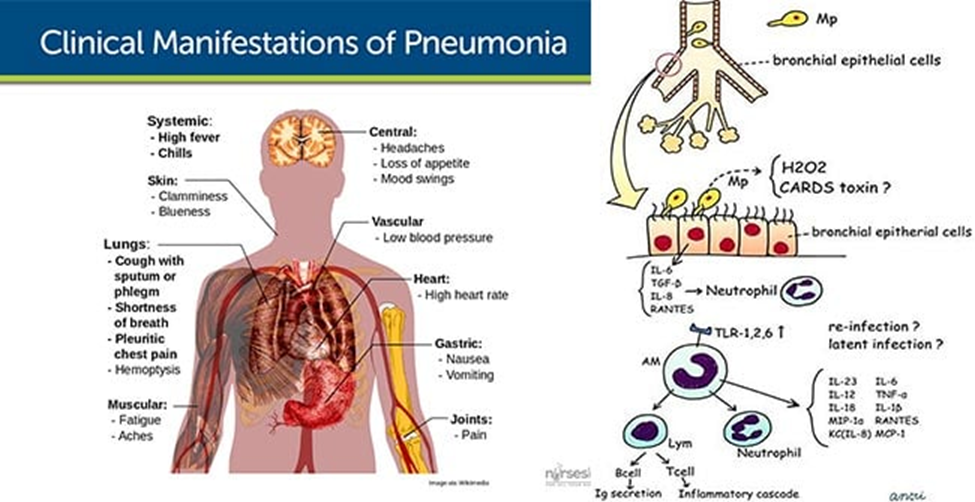
Clinical Manifestations of Mycoplasma pneumoniae
Disease caused: Mycoplasma infection/pneumonia
M. pneumoniae is a prevalent cause of community-acquired pneumonia in healthy individuals younger than 40. The key feature of the diagnosis is the absence of a wet cough. Other diagnoses that can be confused with mycoplasma pneumonia include:
- Aspiration pneumonitis and pneumonia
- Bacterial pneumonia
- Chlamydia pneumoniae
- Coxiella burnetii infection
- Empyema
- Legionella pneumophila
- Lung abscess
- Pediatric pneumonia
- Q fever
- Viral pneumonia
Mycoplasma affects the respiratory tract (throat, windpipe and lungs). Infections are generally mild and it is sometimes called “walking pneumonia”. However, occasionally infections can be severe and need antibiotics.
Symptoms usually appear from 1 to 4 weeks after the person is infected with the bacteria. Symptoms may last for several weeks. Mild infections can be managed safely at home without medicine. Symptoms may include:
- Sore throat
- Dry cough (that may last for weeks or months)
- Headache
- Fever
- Feeling tired
- Shortness of breath
Children younger than 5 years of age may have different symptoms to older children and adults which may include:
- Sneezing
- A stuffy or runny nose
- Watery eyes
- Wheezing
- Vomiting
- Diarrhea
https://microbenotes.com/pathogenesis-and-clinical-manifestations-of-mycoplasma-pneumoniae/
Extra Pulmonary Manifestations:
They are rare and occur either because of active Mycoplasma infection or due to post infectious autoimmune phenomena. Some of the manifestations include:
- Meningoencephalitis, encephalitis, Guilian-Barre syndrome and aseptic meningitis
- Skin rashes including erythema multiforme major (Stevens-Johnson syndrome)
- Myocarditis; pericarditis
- Reactive arthritis
- Hemolytic and hyper coagulopathy
Laboratory Diagnosis of Mycoplasma pneumoniae
Detection of M. pneumoniae infections can be achieved using culture, serology or molecular-based methods. Some reasons that M. pneumoniae detection is not offered by many diagnostic laboratories are that its slow growth and fastidious cultivation requirements make culture impractical. Optimum serological testing requires acute- and convalescent-phase sera collected at least 2 weeks apart, and molecular-based tests are expensive and still have limited availability.
Culture: isolation on specialized media such as Eaton’s agar or PPLO medium; colonies show a characteristic “fried egg” appearance, but growth is slow (1-3 weeks)
Histopathological examination of tissues from persons with acute mycoplasmal respiratory infection, animal models and tracheal organ cultures demonstrate ulceration and destruction of ciliated epithelium of bronchi and bronchioles, edema, bronchiolar and alveolar infiltrates of macrophages, lymphocytes, neutrophils, plasma cells and fibrin. Immunosuppressed people with M. pneumoniae infection may lack pulmonary infiltrates, further attesting to the importance of the host immune response in lesion development.
Serology has historically been the most common laboratory means for diagnosis of M. pneumoniae infections. A fourfold increase in antibody titter in acute and convalescent era is still considered the ‘gold standard’ for the diagnosis of acute M. pneumoniae respiratory infection. M. pneumoniae elicits antibody responses to both lipid and protein antigens, detectable about 1 week after symptom onset, peaking at 3-6 weeks and declining gradually.
Complement fixation (CF) was the earliest serological test used to detect infection by M. pneumoniae primarily measuring the early IgM response but lacking the ability to distinguish between antibody classes. The method has relatively low sensitivity and specificity because the glycolipid antigens used may cross-react with other microorganisms, human tissues, plants. Due to these limitations, CF has largely been replaced by more reliable techniques.
Other serological methods such as immunofluorescent antibody (IFA) assays, hemagglutination tests and particle agglutination assays have been developed but enzyme immunoassays (EIAs) are now the most used commercial tests due to their higher sensitivity and ability to detect antibodies using small serum volumes. EIAs may be qualitative or quantitative and can sometimes achieve sensitivity comparable to PCR if sufficient time has elapsed for antibody development. However, diagnosis often still requires paired acute and convalescent serum samples, which limits rapid diagnosis, although newer rapid point-of-care EIAs detecting IgM and/or IgG have improved clinical detection.
Biochemical Identification
M. pneumoniae is a fastidious organism with limited metabolic capabilities, reflecting its small genome. Key biochemical characteristics and identification features includes:
- Glucose fermentation: the organism ferments glucose producing acid that can be detected in liquid media with phenol red indicator
- Urease negative: does not hydrolyze urea, differentiating it from some other mycoplasma species
- Arginine negative: does not hydrolyze arginine
- Catalase and oxidase negative: lacks these enzymes, consistent with limited oxidative metabolism
- Hydrogen peroxide production generates hydrogen peroxide via a flavin-dependent electron transport chain, contributing to cytotoxicity and use historically for differentiation
Treatment of Mycoplasma pneumoniae
Most patients withM. pneumoniae pneumonia present in outpatient settings and treatment is often with empiric antibiotics for atypical pneumonia.
Macrolides are the first line treatment for M. pneumoniae infection, others include doxycycline or fluoroquinolones. Azithromycin is the most frequently used antibiotic and is usually prescribed for 5 days. Patients receiving doxycycline or fluoroquinolones should be given 7 to 14 days of treatment.
For suspected macrolide-resistant infections, tetracyclines are recommended, such as doxycycline. However, tetracyclines are contraindicated in young children. Fluoroquinolones are another alternative but are contraindicated in children as well.
Prevention and Control of Mycoplasma pneumoniae
Chemoprophylaxis of mycoplasma infections is not recommended and no vaccine is available. Prior natural infection appears to provide the most effective resistance; however, evidence shows that M. pneumoniae infections occur at intervals of several years.
Prevention of M. pneumoniae infection relies on infection control measures and reducing transmission, as no licensed vaccine is currently available. Key strategies include:
- Respiratory hygiene: covering the mouth and nose when coughing or sneezing and proper hand hygiene to reduce spread of respiratory droplets
- Avoiding close contact: limiting exposure in crowded environments such as schools, military barracks, and dormitories during outbreaks
- Prompt diagnosis and treatment: early detection and appropriate antibiotic therapy (e.g. macrolides, tetracyclines or fluoroquinolones) can reduce transmission to others
- Public health measures: isolation of infected individuals during outbreaks and educating the public about respiratory infection control
References
Abdulhadi, B., & Kiel, J. (2025). Mycoplasma pneumonia. In StatPearls [Internet]. StatPearls Publishing.
https://www.ncbi.nlm.nih.gov/books/NBK430780/
Biberfeld, G., & Biberfeld, P. (1970). Ultrastructural features of Mycoplasma pneumoniae. Journal of Bacteriology, 102(3), 855–861.
https://doi.org/10.1128/JB.102.3.855‑861
Bono, M. J. (2024). Mycoplasmal pneumonia: Practice essentials and overview. Medscape. Retrieved June 26, 2024, from
https://emedicine.medscape.com/article/1941994‑overview
Martinez Salazar, M., & Garcia Rodriguez, J. (2021). Clinical aspects of Mycoplasma pneumoniae infections: Pathogenesis and immune response. Pathogens, 10(2), 119. https://www.mdpi.com/2076‑0817/10/2/119
Microbe Notes. (n.d.). Pathogenesis and clinical manifestations of Mycoplasma pneumoniae. Retrieved March 4, 2026, from https://microbenotes.com/pathogenesis‑and‑clinical‑manifestations‑of‑mycoplasma‑pneumoniae/
PathologyOutlines.com. (n.d.). Mycoplasma pneumoniae: Microbiology, pathology, and identification. Retrieved March 3, 2026, from https://www.pathologyoutlines.com/topic/microbiologympneumoniae.html
Razin, S. (1996). Mycoplasmas (Chapter 37). In S. Baron (Ed.), Medical Microbiology (4th ed.). University of Texas Medical Branch at Galveston. https://www.ncbi.nlm.nih.gov/books/NBK7637/
Saraya, T. (2016). The history of Mycoplasma pneumoniae pneumonia. Frontiers in Microbiology, 7, 364.
https://doi.org/10.3389/fmicb.2016.00364
Sleha, R., Bostikova, V., Salavec, M., Bostik, P., Slehova, E., Kukla, R., Mosio, P., Vydrzalova, M., & Mazurova, J. (2013). Mycoplasma infections in humans. Military Medical Science Letters, 4(4), 142–148.
https://doi.org/10.31482/mmsl.2013.023
Ulmeanu, A. I., Ciuparu, G. E., & Matran, E. R. (2025). Characteristics of Mycoplasma pneumoniae pneumonia in Romanian children. Microorganisms, 13(4), 883. https://doi.org/10.3390/microorganisms13040883
Waites, K. B., & Talkington, D. F. (2004). Mycoplasma pneumoniae and its role as a human pathogen. Clinical Microbiology Reviews, 17(4), 697–728. https://doi.org/10.1128/cmr.17.4.697‑728.2004
