Poliovirus remains a key model in virology and vaccinology and a continuing target of global eradication efforts.

- Poliovirus is the etiologic agent of poliomyelitis, a potentially paralytic disease primarily affecting the spinal cord motor neurons, leading to acute flaccid paralysis (AFP) in a small fraction of infections.
- Humans are the only natural host; infection is usually asymptomatic or mild, but historically caused large epidemics of paralysis and death before vaccines became available.
- Since the launch of the Global Polio Eradication Initiative (GPEI) in 1988, wild poliovirus cases have been reduced by >99%, yet eradication is threatened by persistent wild type 1 circulation in Pakistan and Afghanistan and by circulating vaccine-derived polioviruses (cVDPVs).
Poliovirus is also a prototype for understanding RNA virus evolution, mucosal immunity, and the challenges of vaccine-driven eradication in the context of live, attenuated vaccines.
Taxonomy and Classification
- Family: Picornaviridae
- Genus: Enterovirus
- Species: Enterovirus C.
- Serotypes:
- Poliovirus type 1 (PV1)
- Poliovirus type 2 (PV2)
- Poliovirus type 3 (PV3).
- Poliovirus is part of a larger enterovirus group that includes coxsackieviruses and other non-polio enteroviruses, causing meningitis, myocarditis, AFP, and systemic disease.
Key taxonomic features:
- Non-enveloped, positive-sense single-stranded RNA virus with ~7.4 kb genome.
- Classified in Enterovirus C based on genomic sequence relationships, particularly VP1, and biological traits such as host range and receptor usage.
Structure and Morphology
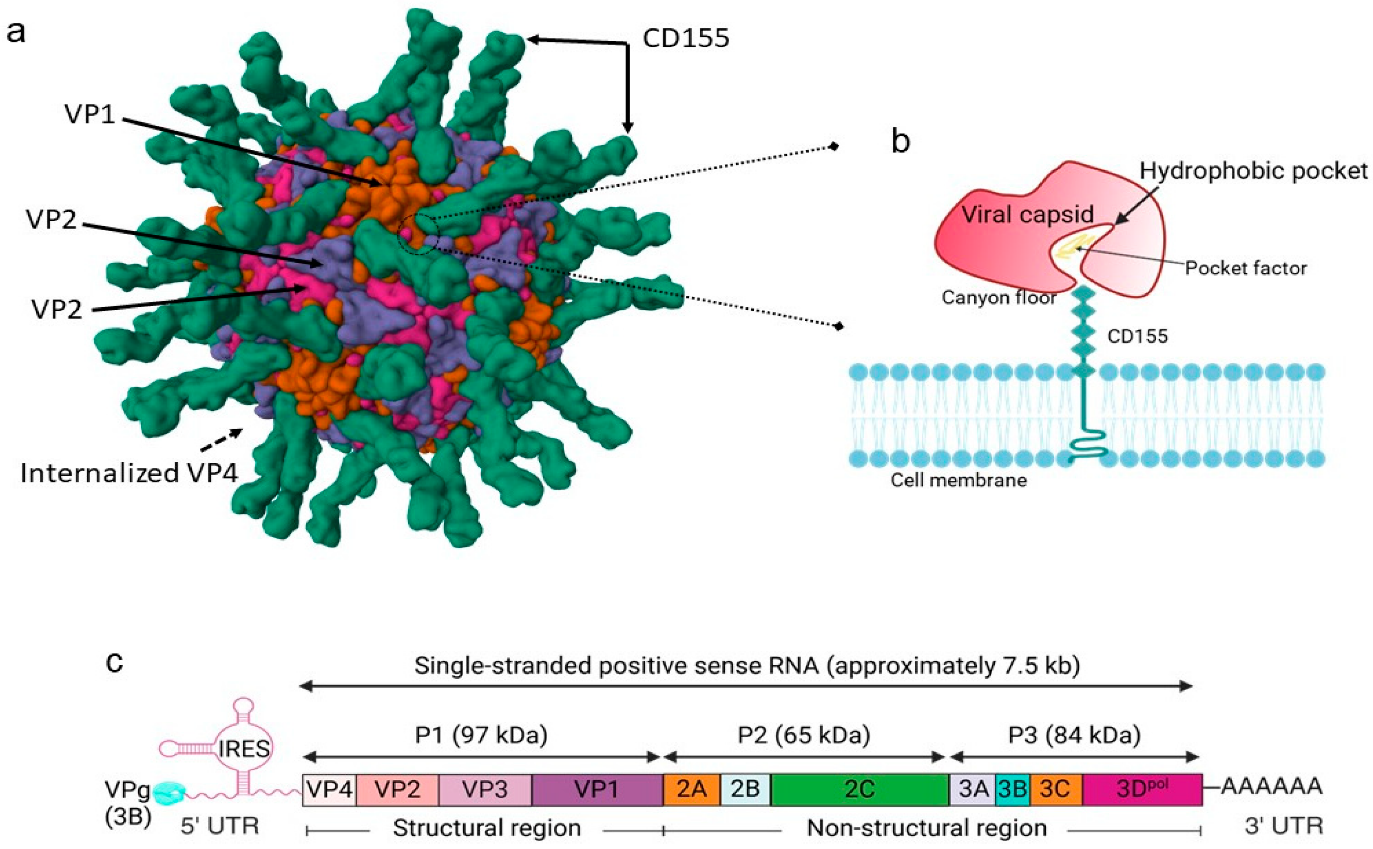
Fig 1: Poliovirus structure and viral genome and polyprotein organization (Xie et al., 2024)
- Capsid architecture:
- Non-enveloped, icosahedral virion ~30 nm diameter.
- Particle composed of 60 copies each of four structural proteins: VP1, VP2, VP3, VP4.
- VP1–VP3 form the outer surface; VP4 is an internal capsid protein lining the inner face of the shell.
- Receptor interaction:
- Host range and tissue tropism are largely determined by capsid proteins and their interaction with the cellular receptor CD155 (poliovirus receptor, PVR).
- Poliovirus binds domain 1 of the extracellular immunoglobulin-like portion of CD155 on intestinal M cells in gut-associated lymphoid tissue (GALT).
- Uncoating mechanism:
- CD155-mediated attachment triggers endocytosis and conformational change of the capsid.
- VP4 externalizes; its hydrophobic N-terminal segment inserts into the endosomal membrane, forming a pore through which the viral RNA is delivered into the cytoplasm.
Genome Organization and Proteins
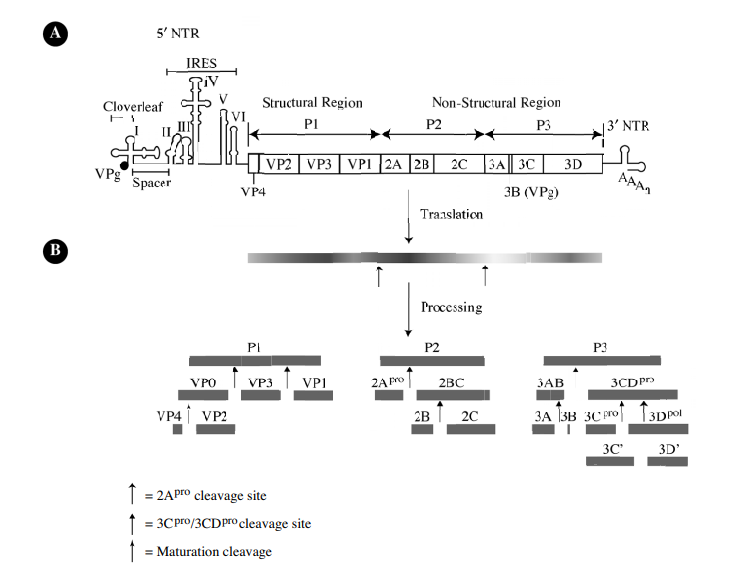
Fig 2: Genomic structure of poliovirus (De Jesus, 2007)
- Genome overview
- Positive-sense, single-stranded RNA, ~7.4 kb in length.
- 5′ end: covalently linked VPg protein; long structured 5′ untranslated region (5′UTR) containing the internal ribosome entry site (IRES).
- 3′ end: short 3′UTR followed by a poly(A) tail.
- Encodes a single large open reading frame (ORF) translated into a polyprotein, post-translational cleaved into structural and non-structural proteins.
- 5′UTR and IRES
- The 5′UTR (~743 nt) has multiple stem-loop domains; domains IV–VI form a type I IRES that recruits ribosomes in a cap-independent manner.
- IRES function relies on canonical initiation factors and IRES-trans-acting factors (ITAFs) plus specific ribosomal proteins (e.g., RACK1) that promote efficient initiation.
- Host proteins like ADAR1 can bind viral RNA and edit adenosines in the IRES, disrupting its structure and significantly reducing translation efficiency.
- Polyprotein and protein groups
- Polyprotein is divided into three regions:
- P1: structural (capsid) proteins VP4, VP2, VP3, VP1.
- P2, P3: non-structural proteins (2A, 2B, 2C, 3A, 3B/VPg, 3C^pro, 3D^pol).
- Cleavage is mediated by viral proteases 2A^pro and 3C^pro/3CD to produce 11–12 mature products.
- Polyprotein is divided into three regions:
- Structural proteins:
- VP1, VP2, VP3: form the outer capsid surface; contain neutralizing antigenic sites that define serotype and are used in molecular epidemiology (particularly VP1 sequencing).
- VP4: internal, myristoylated protein that lines the capsid interior and participates in uncoating and RNA release.
- Changes in the VP1 coding region, including small deletions in key antigenic loops (e.g., BC loop), can modulate neutralization and immune escape while preserving overall fitness.
- Non-structural proteins and key functions
- 2A^pro: cysteine protease that cleaves eIF4G and shuts off host cap-dependent translation, prioritizing IRES-driven viral protein synthesis.
- 2B/2BC: membrane-active proteins that alter intracellular membranes and ion homeostasis, contributing to replication organelle formation.
- 2C: NTPase and helicase-like protein, central in RNA synthesis complexes.
- 3A: membrane protein involved in replication complex anchoring and host trafficking perturbation.
- 3B (VPg): small protein covalently linked to the 5′ end; primer for RNA synthesis and involved in initiation of translation.
- 3C^pro: protease that processes much of the polyprotein; also cleaves host factors, contributing to immune evasion and shutoff.
- 3D^pol: RNA-dependent RNA polymerase with low fidelity, generating high mutation rates and facilitating genetic drift, recombination, and vaccine-derived variants.
- Host RNA editing and restriction
- ADAR1, an adenosine deaminase acting on RNA, edits specific sites in poliovirus RNA:
- Two edits in the IRES → impaired internal initiation of translation.
- One edit in the coding region → amino acid substitution that attenuates replication.
- This illustrates how host RNA-binding proteins can directly shape poliovirus fitness by impacting both translation and protein coding.
- ADAR1, an adenosine deaminase acting on RNA, edits specific sites in poliovirus RNA:
Replication Cycle
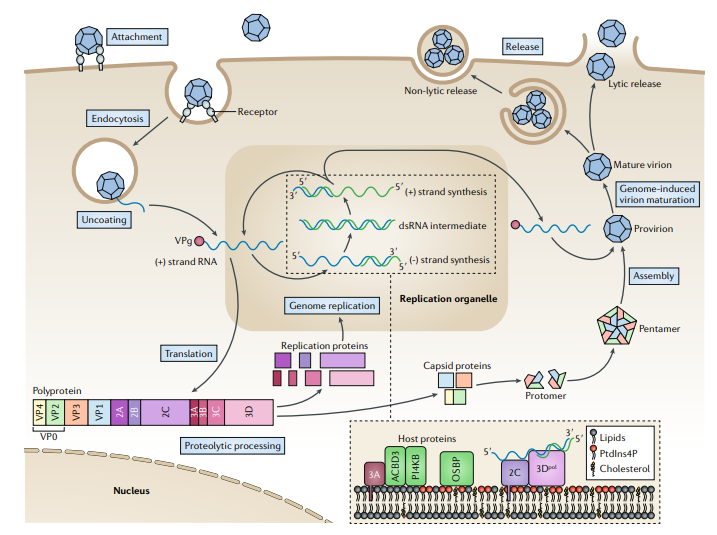
Fig 3: Replication cycle of Poliovirus (Baggen et al., 2018)
- Attachment and entry
- Virus binds to the CD155 (poliovirus receptor, PVR) on susceptible cells, particularly M cells and epithelial cells in the oropharynx and intestinal mucosa.
- Receptor engagement triggers endocytosis of the virion.
- Conformational changes expose VP4 and internalize the RNA into the cytoplasm through pores in the endosomal membrane.
- Uncoating and early translation
- Poliovirus RNA, already in mRNA sense, is released directly into the cytoplasm and immediately translated.
- Translation initiation is cap-independent, driven by the IRES in the 5′UTR.
- Specific ribosomal proteins (e.g., RACK1) enhance IRES-mediated initiation; knockout of RACK1 significantly reduces viral protein synthesis and delays host translation shutoff.
- Polyprotein processing and host shutoff
- The entire ORF is translated into a single polyprotein that is co– and post-translational cleaved by viral proteases.
- Viral 2A and 3C proteases cleave host initiation factor eIF4G and poly(A)-binding protein, shutting down cellular cap-dependent mRNA translation while leaving viral IRES-dependent translation active.
- This selective translational takeover is central to poliovirus replication and pathogenicity.
- Replication complex formation
- Non-structural proteins remodel intracellular membranes (ER, Golgi-derived vesicles) into replication organelles.
- 3D^pol synthesizes a complementary negative-strand RNA, which serves as a template for multiple positive-strand genomes.
- Replication is tightly associated with VPg priming and host factors; ADAR1 and other RBPs can bind replicating RNA and modulate output.
- Genome amplification and quasispecies
- Error-prone 3D^pol results in a swarm of closely related genomes (quasispecies), enabling rapid adaptation and emergence of vaccine-derived polioviruses (VDPVs) or neutralization escape variants.
- Recombination with other Enterovirus C strains further diversifies genomes.
- Assembly and maturation
- Structural proteins assemble into protomers → pentamers → provirions that package new positive-strand genomes.
- Maturation involves conformational rearrangements, VP0 cleavage into VP2 and VP4, and stabilization of the capsid.
- Release
- Poliovirus is non-enveloped and typically exits cells via cell lysis, releasing high titers of virions into the intestinal lumen and bloodstream, seeding further infection.
Pathogenesis and Host Immune Response
- Initial infection and primary replication
- Entry via the fecal–oral route; initial replication in:
- Oropharyngeal mucosa.
- Intestinal epithelial cells and gut-associated lymphoid tissue (GALT).
- Most infections are asymptomatic or cause minor, non-specific illness, with the virus confined to the gut and regional lymph nodes.
- Entry via the fecal–oral route; initial replication in:
- Viremia and neuroinvasion
- After primary replication, the virus can enter the bloodstream (primary and sometimes secondary viremia).
- Poliovirus shows a tropism for motor neurons of the anterior horn of the spinal cord, brainstem, and motor cortex.
- Neuroinvasion routes (from contemporary reviews):
- Hematogenous spread across the blood–brain barrier or blood–CSF barrier.
- Retrograde axonal transport via peripheral motor nerves.
- Once in the CNS, poliovirus replicates in motor neurons, leading to cell lysis, inflammation, and neuronal death.
- Neuropathology
- Pathological hallmarks:
- Lymphocytic infiltrates in the spinal cord gray matter.
- Degeneration and loss of anterior horn cells, especially in the lumbar and cervical cord.
- Lesions can extend to bulbar motor nuclei and higher centers, causing bulbar and polio encephalitic forms.
- Pathological hallmarks:
- Acute immune response
- Innate immunity:
- Type I interferons (IFN-α/β) and innate sensing limit early replication; enteroviruses, including poliovirus, encode proteins (2A, 3C, 3A) that antagonize IFN signaling and host RNA metabolism (described broadly for enteroviruses).
- ADAR1 acts as an intrinsic antiviral factor by editing poliovirus RNA, reducing IRES function, and introducing attenuating mutations.
- Adaptive immunity:
- Serum neutralizing IgG is the primary correlate of protection against paralytic disease; it prevents viremic spread to the CNS.
- Secretory IgA in the gut is critical to limit replication and shedding; strong after OPV, weaker after IPV alone (shown in recent mucosal immunity work).
- Innate immunity:
- Immune evasion strategies
- Rapid replication and high mutation rate generate escape variants from neutralizing antibodies, particularly under suboptimal immunity.
- Disruption of host protein synthesis (eIF4G and PABP cleavage) blunts host antiviral protein production.
- Use of specialized ribosomal proteins (e.g., RACK1) and IRES allows poliovirus translation to proceed when host translation is largely shut down.
- Long-term sequelae: Post-polio syndrome (PPS)
- Occurs decades after acute poliomyelitis in survivors.
- Characterized by:
- New or progressive muscle weakness in previously affected or seemingly unaffected muscles.
- Fatigue, muscle and joint pain, and functional decline.
- Pathophysiology (from clinical review):
- Chronic overwork of surviving motor neurons that had sprouted to reinnervate denervated fibers.
- Gradual loss of these enlarged motor units, leading to renewed denervation and symptoms.
Epidemiology and Transmission
- Transmission:
- Predominantly faecal–oral, via direct contact, contaminated water or food; respiratory secretions may occasionally contribute.
- Primary replication in the gut leads to high‐titer shedding in stool, enabling environmental spread; poliovirus is frequently detected in sewage and environmental surveillance.
- Global epidemiology:
- Before vaccines, wild polioviruses caused >500,000 cases of paralysis or death annually worldwide.
- As of the 2020s, wild poliovirus type 1 (WPV1) transmission persists mainly in Pakistan and Afghanistan, with occasional importations causing outbreaks in previously polio-free regions.
- Wild poliovirus types 2 and 3 are certified eradicated, but cVDPV2 and cVDPV3 pose major challenges, causing more paralytic cases globally than WPV in recent years.
- Vaccine-derived poliovirus (VDPV):
- OPV strains can revert or recombine with other Enterovirus C strains after replication in under-immunized populations, regaining neurovirulence and transmissibility.
- Between Jan 2018 and May 2023, cVDPV-linked cases (3,442) outnumbered WPV-linked cases (390) by ~8.8:1.
- 2022 London sewage detection of type 2 VDPV lineage highlighted silent transmission in high-income settings using IPV-only schedules.
Clinical Manifestations
- Spectrum of clinical outcomes (only a minority develop severe disease):
- Inapparent infection: up to 90–95% of infections; asymptomatic, but virus may still be shed.
- Abortive poliomyelitis (minor illness):
- Nonspecific febrile illness with malaise, anorexia, headache, sore throat, and sometimes mild GI symptoms.
- No CNS involvement, full recovery.
- Non-paralytic poliomyelitis (aseptic meningitis):
- More intense fever, headache, neck and back stiffness, photophobia.
- CSF pleocytosis; symptoms resolve without permanent paralysis.
- Paralytic poliomyelitis
- Occurs in <1% of infections, but is responsible for classical polio morbidity.
- Clinical phases (from neurological descriptions):
- Pre-paralytic phase:
- Non-specific systemic symptoms ± meningeal signs.
- Myalgia and muscle tenderness may be prominent.
- Paralytic phase:
- Rapid onset (often over hours) of asymmetric flaccid paralysis; legs are commonly affected more than arms.
- Loss of deep tendon reflexes in affected limbs; sensory function is typically preserved.
- Muscle atrophy follows over weeks to months as motor neurons die.
- Bulbar poliomyelitis:
- Involvement of cranial nerve nuclei → dysphagia, dysarthria, facial weakness.
- Respiratory failure due to diaphragmatic and intercostal muscle paralysis; historically required “iron lung” support.
- Polio encephalitis:
- Less common; can present with confusion, seizures, or other cortical signs.
- Pre-paralytic phase:
- Determinants of severity
- Higher risk of paralytic disease and severe outcomes in:
- Unvaccinated individuals.
- Adults compared with young children.
- Pregnant women, individuals with tonsillectomy, or intense physical exertion around the onset (classically associated with a higher risk).
- Viral factors:
- Neurovirulence is dictated largely by capsid determinants (e.g., in VP1) and 5′UTR/IRES elements that influence tropism and replication efficiency in neuronal cells.
- Higher risk of paralytic disease and severe outcomes in:
- Complications and long-term consequences
- Acute: respiratory failure, autonomic disturbances, rhabdomyolysis, and secondary infections.
- Chronic: residual paralysis, limb deformities, scoliosis, chronic pain, and post-polio syndrome years later.
Laboratory Diagnosis
- Sample types:
- Stool samples are the primary specimen for suspected AFP.
- Throat swabs and cerebrospinal fluid (CSF) may be useful but are less sensitive.
- Environmental surveillance uses sewage and wastewater samples.
- Virus isolation (WHO standard):
- Parallel inoculation into:
- L20B cells (murine cell line expressing human CD155) – selectively grows poliovirus.
- RD (human rhabdomyosarcoma) cells – support a broad range of enteroviruses.
- Viruses that grow in RD but not L20B are classified as non-polio enteroviruses and generally not further characterized.
- Parallel inoculation into:
- Molecular typing:
Key Diagnostic Approaches for Poliovirus
Table 1: Key laboratory methods for detecting and characterizing poliovirus.
| Step / Method | Purpose / Notes |
| L20B and RD cell culture | Initial isolation, poliovirus vs. non-polio enteroviruses |
| qPCR serotyping, intra-typic testing | Distinguish types 1–3, vaccine vs. non-vaccine strains. |
| VP1 and whole-genome sequencing | Track origins, monitor evolution, define VDPV lineages |
| Direct PCR + NGS on stool/sewage | Faster detection without culture (~2 days vs. 2–3 weeks) |
- Genetic sequencing underpins phylogenetics, outbreak tracing, and cVDPV detection, and is central to modern eradication programs.
Treatment and Antiviral Therapy
- There is no specific, widely used antiviral therapy for poliovirus; management of paralytic disease is supportive (respiratory support, prevention of complications).
- For enteroviruses generally, research on direct-acting antivirals (capsid binders, protease and polymerase inhibitors) and host-targeting agents is advancing, but no polio-specific drugs are licensed.
- Experimental approaches (e.g., broad-spectrum enterovirus antivirals) may have theoretical activity against poliovirus, but current control depends almost entirely on vaccination and public health measures.
Prevention and Control
Recent literature emphasizes that vaccination is the cornerstone of prevention, with evolving strategies to balance efficacy, intestinal immunity, and genetic stability.
Inactivated Poliovirus Vaccine (IPV)
- First used in 1955; currently the mainstay in many high-income and some middle-income countries.
- Intramuscular or subcutaneous injection of formalin-inactivated viruses of all three serotypes.
- Key features:
- Induces strong systemic humoral immunity; excellent protection from paralytic disease.
- Historically thought to induce limited intestinal immunity; newer data suggest some boosting of mucosal responses, particularly when combined with OPV.
- Requires stringent biocontainment during manufacture due to large volumes of live virus used.
- More expensive and logistically complex to deliver than OPV (trained personnel, injection equipment).
- Recent developments:
- Efforts toward dose-sparing intradermal IPV, novel production platforms, and non-infectious VLP-based IPV to reduce facility risk and cost.
Oral Poliovirus Vaccine (OPV)
- Live attenuated Sabin strains, historically trivalent (tOPV), are now often bivalently attenuated (bOPV, types 1 and 3) due to global withdrawal of Sabin 2.
- Advantages:
- Induces robust intestinal IgA and blocks virus shedding, key for interrupting transmission.
- Easy and inexpensive to deliver orally, enabling mass campaigns.
- Major challenge:
- Genetic instability of Sabin strains; reversion and recombination can yield cVDPV with restored neurovirulence and enhanced transmission.
Novel OPVs and Next-Generation Vaccines
- nOPV2 (novel type 2 OPV):
- Rationally engineered for increased genetic stability while retaining immunogenicity; authorized under WHO Emergency Use Listing for cVDPV2 outbreaks.
- Early data indicate reduced reversion, good immunogenicity, and lower risk of seeding new cVDPV2 lineages.
- nOPV1 and nOPV3:
- New candidates generated by swapping capsid regions of nOPV2 with Sabin 1 or 3 capsid coding sequences.
- More attenuated yet immunogenic in animal models; show enhanced genetic stability under accelerated evolution experiments.
- Research pipeline:
- Virus-like particle (VLP) vaccines, mRNA-based candidates, and micro-needle patch delivery are under investigation to improve safety, ease of administration, and mucosal immunity.
Mucosal Immunity and Vaccine Schedules
- Clinical trials and meta-analyses show that different combinations of IPV and OPV yield distinct patterns of stool shedding after live virus challenge, reflecting varying degrees of intestinal immunity.
- Enteric neutralizing IgA in stool correlates with reduced magnitude and duration of shedding.
- Sequential IPV–OPV schedules can modulate shedding kinetics and genetic diversity of shed OPV strains, informing post-eradication risk management.
Surveillance and Public Health Strategies
- AFP surveillance in children, combined with environmental sewage surveillance and high-throughput sequencing, is central to early detection of circulation, including in IPV-only countries.
- Mathematical modeling plays a major role in shaping eradication policy, evaluating trade-offs among vaccine use, cVDPV risk, and cost-effectiveness.
- Current WHO and GPEI strategies emphasize:
- Maintaining high population immunity with context-appropriate IPV/OPV use.
- Rapid outbreak response using targeted OPV (including nOPV2) and/or IPV campaigns.
- Continued environmental surveillance and genetic sequencing to detect silent transmission.
- Long-term planning for cessation of OPV once all wild and vaccine-derived viruses are eliminated, shifting to IPV-based maintenance and stringent biocontainment.
Conclusion
Poliovirus is a small, non-enveloped enterovirus with a positive-sense RNA genome and strict human tropism, transmitted mainly by the faecal–oral route and only rarely invading the CNS to cause paralytic poliomyelitis. Its biology, especially intestinal replication, mucosal immunity, and high mutation rate, has shaped both its pathogenesis and the complex landscape of vaccine design and eradication policy. While wild poliovirus has been driven to the brink of eradication, the emergence and spread of cVDPVs underscore the dual nature of live attenuated vaccines and the need for more genetically stable formulations, expanded IPV use, and robust environmental and genetic surveillance. Recent advances in molecular virology, structural vaccinology, and modeling offer realistic paths toward a polio-free world, but success will depend on sustained immunization coverage, innovation in vaccine platforms, and continued global commitment.
References
Baggen, J., Thibaut, H. J., Strating, J. R. P. M., & Van Kuppeveld, F. J. M. (2018). The life cycle of non-polio enteroviruses and how to target it. Nature Reviews Microbiology, 16(6), 368–381. https://doi.org/10.1038/s41579-018-0005-4
Devaux, C., Pontarotti, P., Levasseur, A., Colson, P., & Raoult, D. (2024). Is it time to switch to a formulation other than the live attenuated poliovirus vaccine to prevent poliomyelitis? Frontiers in Public Health, 11, 1332501.
Jartti, M., Flodström-Tullberg, M., & Hankaniemi, M. M. (2024). Enteroviruses: Epidemic potential, challenges, and opportunities with vaccines. Journal of Biomedical Science, 31, 63.
Jones, J. E., Le Sage, V., & Lakdawala, S. S. (2020). Viral and host heterogeneity and their effects on the viral life cycle. Nature Reviews Microbiology, 18(12), 709–725.
Jorgensen, D., Pons-Salort, M., Shaw, A. G., & Grassly, N. C. (2020). The role of genetic sequencing and analysis in the polio eradication programme. Virus Evolution, 6(1), veaa040.
Bandyopadhyay, A., & Macklin, G. R. (2020). Final frontiers of the polio eradication endgame. Current Opinion in Infectious Diseases, 33(5), 372–380.
Kim, C. Y., Piamonte, B., Allen, R., & Thakur, K. T. (2023). Threat of resurgence or hope for global eradication of poliovirus? Current Opinion in Neurology, 36(4), 520–528.
Thompson, K. M., & Kalkowska, D. A. (2020). Review of poliovirus modeling performed from 2000 to 2019 to support global polio eradication. Expert Review of Vaccines, 19(8), 661–682.
Liang, J., Zhang, Q., Li, Y., & Wang, L. (2025). Advances and challenges in poliomyelitis vaccines: A comprehensive review of development, production, and global deployment. Frontiers in Public Health.
Yeh, M.-T., Smith, M. M., Carlyle, S., Konopka-Anstadt, J. L., Burns, C. C., Konz, J. O., Andino, R., & Macadam, A. J. (2023). Genetic stabilization of attenuated oral vaccines against poliovirus types 1 and 3. Nature, 618(7966), 767–774.
Singh, S., Mane, S., Kasniya, G., Cartaya, S., Rahman, M. M., Maheshwari, A., Motta, M., & Dudeja, P. (2022). Enteroviral infections in infants. Newborn, 1(3), 246–261.
Sutter, R. W., Eisenhauer, M., Molodecky, N. A., Verma, H., & Okayasu, H. (2024). Inactivated poliovirus vaccine: Recent developments and the tortuous path to global acceptance. Pathogens, 13(3), 218.
Klapsa, D., Wilton, T., Zealand, A., Bujáki, E., Saxentoff, E. V., Troman, C., Shaw, A. G., Tedcastle, A., Majumdar, M., Mate, R., Akello, J., Huseynov, S., Zeb, A., Zambon, M., Bell, A., Hagan, J. E., Wade, M. J., Ramsay, M., Grassly, N. C., Saliba, V., & Martin, J. (2022). Sustained detection of type 2 poliovirus in London sewage between February and July, 2022, by enhanced environmental surveillance. The Lancet, 400(10363), 1067–1076.
Connor, R. I., Brickley, E. B., Wieland-Alter, W. F., Ackerman, M. E., Weiner, J. A., Modlin, J. F., Bandyopadhyay, A. S., & Wright, P. F. (2021). Mucosal immunity to poliovirus. Mucosal Immunology, 14(6), 1223–1237.
Fu, Y.-G., Ma, R., Zhao, Z., Mo, Z., Ying, Z., Li, J., Ye, H., Li, G.-L., Liu, X., Liang, J., Ping, L., Li, J., Tao, J.-H., Yang, Q., Wei, D.-K., Yi, L., Chen, H.-B., Wang, J.-F., Jiang, R., Yu, L., Cai, W., Yang, W., Yue, L., Xie, M., Yin, Q., Pu, J., Hong, C., Cai, L.-K., Deng, Y., Wen, J., Ma, Y., Gao, N., Wang, X., Liao, H., Ji, Q., Ji, G., Hu, W., Gu, Q., He, X., Chu, H., Fu, Y., Zhou, J., Wen, Y., Yang, X., Li, C.-G., Shi, L., Zhao, T., Huang, T., & Yang, J.-S. (2025). Poliovirus shedding after sequential immunization of Sabin-strain inactivated polio vaccines and oral attenuated polio vaccines. NPJ Vaccines, 10, 18.
Lee, M. F., Tham, S. K., & Poh, C. L. (2025). Antiviral strategies targeting enteroviruses: Current advances and future directions. Viruses, 17(9), 1845.
Liu, Y., Maisimu, M., Ge, Z., Xiao, S., & Wang, H. (2025). The pathogenesis and virulence of the major enterovirus pathogens associated with severe clinical manifestations: A comprehensive review. Cells, 14(10), 1234.
Xie, H., Rhoden, E. E., Liu, H., Ogunsemowo, F., Mainou, B. A., Burke, R. M., & Burns, C. C. (2024). Antiviral development for the polio endgame: current progress and future directions. Pathogens, 13(11), 969. https://doi.org/10.3390/pathogens13110969
