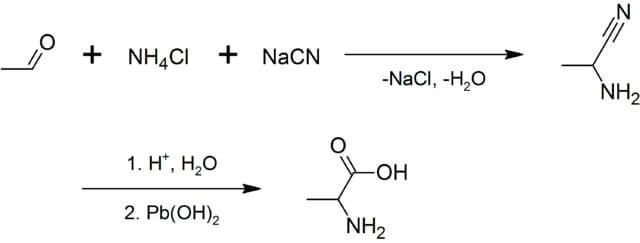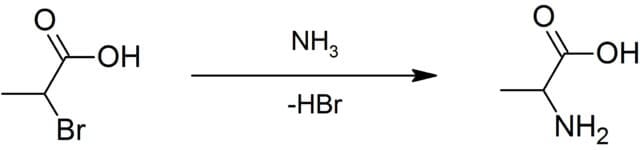
What is Alanine?
A group of organic compounds that contains amino and carboxyl functional groups is amino acids. If carbon atoms are attached to carboxyl and amino groups then the amino acids are referred to as α-amino acids. Amino acids are amphoteric as they contain both acidic and basic groups and are soluble in water. Amino acids are classified in different ways according to their structure, chemical characteristics, nutritional requirements, and metabolic reactions. Amino acids that are classified based on their structure are divided into seven groups. Twenty amino acids are divided into seven groups based on their structure. Alanine, Glycine, Valine, Leucine, and Isoleucine have aliphatic side chains. Amino acids that have aliphatic side chains are mono-amino monocarboxylic acids. Amino acids show properties and reactions similar to both amines and carboxylic acids. Twenty amino acids are considered as standard amino acids of protein as they are found nearly in all proteins.
Definition of Alanine
Alanine is an aliphatic amino acid that has two amino acids; L-alanine or α-alanine is a constituent of proteins. Alanine was isolated in 1879 from silk fibroin which is a rich source of L-alanine. Alanine is considered non-essential amino acid because mammals and birds can synthesize from pyruvic acid that is formed after the breakdown of carbohydrates. Carnosine and anserine are two peptides that are the house for D-alanine or β-alanine. Alanine is α-amino acid that is the analog of α-keto acid pyruvate. β-alanine is used in plants and microorganisms in the synthesis of pantothenic acid or vitamin B5 also is a component of coenzyme A.
Structure of Alanine
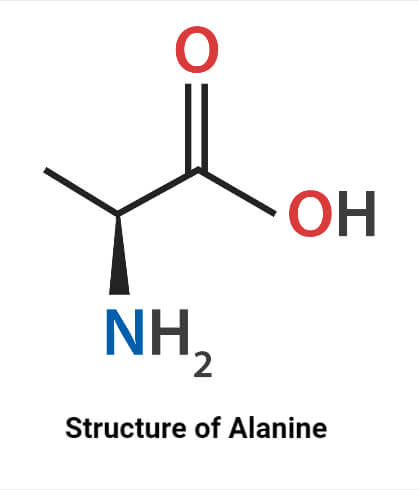
Its molecular formula is C3H7NO2 that is abbreviated as Ala. Alanine is an alpha-amino propionic hydrophobic amino acid having an alkyl group in the side chain. They are glucogenic according to their metabolic properties. It contains carbon on the center that is attached to both the amine and carboxylic groups. They have one amino and one carboxylic group in their structure so considered simple amino acids. They are hydrophobic and have no charge in the R group. Beta carbon is achiral whereas larger amino acids have chiral atoms. Alanine has a side chain that doesn’t bind with a hydrogen bond. The methyl group present in it is non-reactive and not involved in protein function directly. The Amine group is protonated and the carboxyl group is deprotonated in its zwitterionic forms.
Sources of Alanine
As it is a non-essential amino acid, the body can directly produce it but is mainly found in meat products and also through dairy products. Animal sources: seafood, eggs, fish, meat, dairy products. Plant sources: Beans, nuts, corn, soybeans, brown rice, legumes.
Physical Properties of Alanine
- Non-polar, Uncharged
- Orthorhombic crystals from water.
- Sweet in taste
- Odorless
- Soluble in water, acids, and alkalis.
- Insoluble in organic solvents.
- Hydrophobic
- Ambivalent
- Colorless
- Levorotatory at pH 7.0
Chemical Properties of Alanine
- The reaction of amino and carboxyl group. Alpha-keto acid is formed in the reaction due to amino group.
- Has an L configuration and contains an asymmetric carbon atom.
- Boiling point: 250ºC
- Melting point: 300ºC
- Isoelectric point: 6.0
- pKa: -12
- Zwitterionic form of L-alanine at pH 7.3
Biosynthesis of Alanine
Image Source: Organic Syntheses, Inc.
Alanine can be synthesized from different components like pyruvic acid, serine, tyrosine, aspartic acid, and cysteine. It occurs in plasma in its free state at high levels. Alanine is synthesized from pyruvate and branched-chain amino acid that plays important role in the glucose-alanine cycle. This cycle enables pyruvate and glutamate to be removed muscle and safely transported to the liver. In muscle, pyruvate is converted to alanine while in the liver alanine is converted to pyruvate. Mainly, the synthesis of alanine occurs when fasting or had no food for a long time. The formation of pyruvic acid involves two main important steps; transamination and oxidative deamination. Transamination requires alpha-amino acid that generates alpha-keto acid in the process. Transamination of pyruvate is a major path in the synthesis of alanine. AvtA and AvtB are two alanine-forming transaminases that catalyze the reaction. Then, alanine is transported to the liver by the process of Gluconeogenesis, glucose is formed from pyruvic acid. This form of energy produced by the body is called the Glucose-Alanine cycle that is used as fuel that is released from muscles. d-alanine is synthesized from L-alanine with the help of racemase which is then degraded to pyruvate and ammonia by non-specific membrane-bound d-amino acid dehydrogenase.
Functions and Uses of Alanine
- It is an amino acid that is used to make proteins.
- It may help to treat diabetes as it helps the body use glucose.
- It helps prevent hypoglycemia.
- It is used to break down tryptophan and vitamin B-6 as an energy source for Central Nervous System and muscles.
- It also helps in strengthening the immune system by producing antibodies.
- It is required for the breakdown of glucose.
- Alanine also helps the body to use sugar for metabolism.
Safety and Toxicity of Alanine
- Intake of amino acids leads to negative nitrogen balance.
- In children, it may affect the physical growth of the body.
- Alteration in the alanine cycle increases the level of alanine transferase enzyme that results in the development of diabetes.
- Neurotoxicity like seizures and sleep disorders.
- Respiratory distress
- A high level of alanine leads to higher blood pressure.
Frequently Asked Questions (FAQs)
Question- Alanine is polar or nonpolar?
Answer- Alanine is a nonpolar aliphatic amino acid.
Question- What foods are high in alanine?
Answer- Animal sources: seafood, eggs, fish, meat, dairy products. Plant sources: Beans, nuts, corn, soybeans, brown rice, legumes.
References
- Acids A. (n.d.). Amino acids, peptides, and proteins 24-1.
- From, https://www.britannica.com/science/alanine
- From, https://go.drugbank.com/drugs/DB00160
- From, https://pubchem.ncbi.nlm.nih.gov/compound/Alanine#section=Color-Form
- From, https://www.urmc.rochester.edu/encyclopedia/content.aspx?contenttypeid=19&contentid=Alanine
- Lehninger A L, Nelson D L and Cox M M (2000). Lehninger principles of biochemistry. New York: Worth Publishers.
- Satyanarayana U and Chakrapani U (2013). Biochemistry. Elsevier. Pp: 44-47.
- Synthesis A A, Cycles, C, Biosynthesis, P G., Processes, E T., Biosynthesis, G., Cycle G, Control, T. (n.d.). No Title.