All matter is composed of atoms that can combine to form elements, compounds, and molecules at the molecular level. Despite having so many different parts, matter can be broadly divided into two types based on its composition: pure substances and mixtures.

Pure Substances
Pure substances are made up of only one type of particle, the atomic kind. They have an unchanging structure. Pure substances can be further divided into elements and compounds.
Elements: A pure substance made up of just one kind of atom is referred to as an element. There are no physical or chemical processes that can break it down or change it into new substances. Most elements fall into one of three categories: metals, non-metals, or metalloids.
Compounds: When two or more elements are mixed chemically in a predetermined fixed ratio, the result is a pure substance known as a compound. Chemical processes can be used to break down and change these chemicals into new ones. Consider the gas carbon dioxide.
Properties of Pure Substance
- Pure substances typically only include one type of atom or molecule and are homogeneous in nature.
- The majority of these substances have a constant or homogenous makeup.
- The boiling and melting points of the substances are constant.
- In a chemical reaction, a pure substance often takes part to produce predictable results.
Formula Unit
An elemental composition specifies which and how many atoms make up a chemical substance. Since compounds are made up of a particular ratio of various elements, there must be a word to describe how they are held together in predetermined ratios. The smallest whole number ratio of atoms that can be used to characterize a chemical is represented by a formula unit. In other words, it is the smallest group of atoms from which the chemical formula of a substance may be deduced.
The formula unit for table salt is 1:1 i.e. there is one chloride ion for every sodium ion.
Instead of focusing on the specific particles that make up a compound, formula units can help us examine the relative amounts of the various constituents.
Percentage Composition
When analyzing a compound, it’s common to want to know how all the different elements in the compound compare to the compound as a whole. Check out the Law of Definite Proportions for a more in-depth look at this idea!
The percentage composition reveals the mass percentage of each individual element in the compound.
The formula for percent composition is:

Calculating Percentage Composition
- The atomic masses of elements must be added using the periodic table to calculate the molecular mass of the given compound.
- Now multiply the mass by that amount if there are many atoms.
- Now divide each atom’s mass by the compound’s total molecular mass.
For Example:
- Calculate the Percentage composition of NaCl.
The mass of Na = 22.99 g/mol
The mass of Cl = 35.45 g/mol
Sum of Atomic Mass of Na and Cl = (22.99 + 35.45) g/mol = 58.44 g/mol
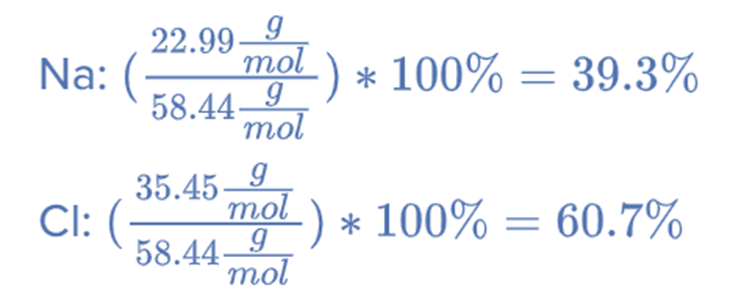
Therefore, NaCl is made up of 39.3% Na and 60.7% Cl.
- Calculate percent composition of potassium in K2Cr2O7.
The molecular weight of K2Cr2O7:
2(39 g/mol) + 2(52 g/mol) + 7(16 g/mol) = 294 g/mol.
Out of this total weight, the percent composition of K:
(2(39 g/mol)/294 g) = 26.5%
Empirical Formula
Information about the ratio of atom numbers in the compound is provided by the empirical formula. The percent composition of a compound directly leads to its empirical formula. The simplest whole-number ratio of the various atoms in a compound is represented by an empirical formula.
Consider glucose as an example. The empirical formula for glucose is CH2O, while its molecular formula is C6H12O6.
Watch out the video for some calculation examples of the elemental composition of pure substances.
References
- Brown, Theodore L, H E. LeMay, Bruce E. Bursten, Catherine J. Murphy, Patrick M. Woodward, and Matthew Stoltzfus. Chemistry: The Central Science. , 2015. Print.
- Fraser, Simon. Classifying Matter According to Its Composition, LibreTexts: Chemistry, 2020.
- https://www.iitianacademy.com/ap-chemistry-1-3-elemental-composition-of-pure-substances-exam-style-questions-with-answer-mcq/
- https://www.studysmarter.co.uk/explanations/chemistry/physical-chemistry/elemental-composition-of-pure-substances/
