Flerovium is a synthetic transition metal with an atomic number of 114 and is represented by the symbol ‘Fl’ in the periodic table. It is silvery in appearance and belongs to the p-block of period 7 of the periodic table. Only tiny quantities of Flerovium have been successfully synthesized, hence there isn’t much known about it based on experimental data, but some qualities can be predicted using periodic table trends.
Flerovium, an extremely radioactive element that does not occur naturally, and is produced inside a laboratory setting and decays within milliseconds after being synthesized. This element, previously known as ununquadium, was formally named in 2012 after the Flerov Laboratory of Nuclear Reactions (FLNR) of the Joint Institute for Nuclear Research in Dubna, Russia, where just a single atom of this element was initially synthesized in 1998. Flerovium has been designated after physicist Georgy Flerov, who founded the Joint Institute of Nuclear Research (JINR).
History and Discovery of Flerovium
- Dmitri Mendeleev anticipated the presence of element 114, which was dubbed eka-lead.
- In the year 1999, a group of physicists supervised by Yuri Oganessian at the Joint Institute of Nuclear Research (JINR) in Dubna, Russia, created Flerovium.
- They blasted plutonium-244 with calcium-48 nuclei, yielding one atom of flerovium-289.
- Long before its discovery, flerovium isotopes had been predicted to be found in the island of stability (elements ranging from copernicium (112) to oganesson (118)) and to have a magic number of protons and neutrons of 114 protons and 184 neutrons, following helium-4, oxygen-16, calcium-48, and lead-208, the other members of the doubly magic atomic numbers group.
- It has been postulated that the nuclei flerovium would be oblate and distorted, resulting in resistance to spontaneous fission.
- It required 5 billion atoms of calcium to generate one atom of flerovium, and the experiment lasted 40 days.
- In 1972, it was proposed that flerovium-298 have a half-life of approximately one year.
- The element Flerovium got its name after physicist Georgy Flerov, who founded the Joint Institute of Nuclear Research (JINR).
Occurrence of Flerovium
- Flerovium can be synthesized artificially. It’s a synthetic element that is extremely unstable. Its half-life is only a few seconds.
- Flerovium is a synthetic radioactive metal formed by nuclear bombardment and has only been manufactured in trace amounts.
- Flerovium is only found in specialized laboratories due to its rapid decay.
- The most recent tests employed the GSI linear accelerator to fire four trillion calcium-48 ions at 10% the speed of light per second against a plutonium-244 target. As a result, a handful of flerovium atoms were generated every day.
- Despite a slow start in synthesis, approximately 80 atoms have been observed until now, 50 of which were purposefully synthesized and the remaining 30 from radioactive decay of heavier elements.
- Flerovium is formed by blasting 244Pu with 48Ca in a heavy ion accelerator.
- Flerovium has five isotopes having known half-lives, ranging in mass from 285 to 289.
Elemental Properties of Flerovium
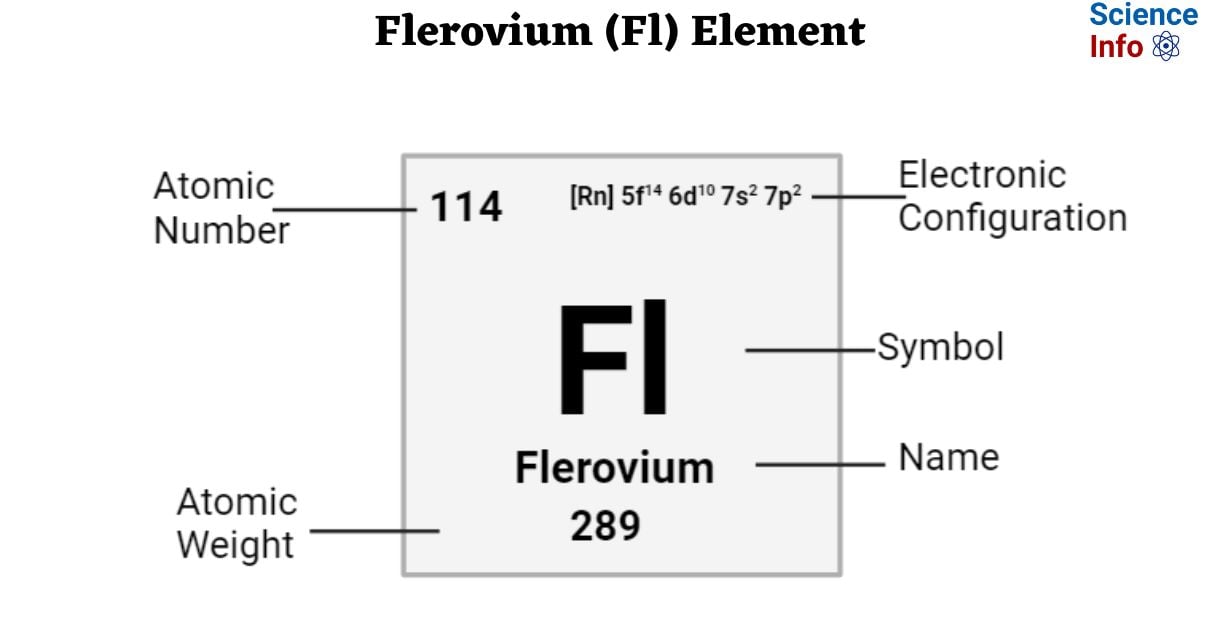
| Electronic Configuration | [Rn] 5f14 6d10 7s2 7p2 |
| Atomic Number | 114 |
| Atomic Weight | 289 g.mol -1 |
| State at 20°C | Solid |
| Group, Period, and Block | Trans-actinides, 7, p-block |
| Density | 14 g/cm3 (estimated) |
| Ionic radius | – |
| Van der Waals radius | – |
| Electron shells | 2, 8, 18, 32, 32, 18, 4 (estimated) |
| Electrons | 114 |
| Protons | 114 |
| Neutrons | 175 |
Isotopic Information of Flerovium
- Flerovium has no stable isotopes naturally, but they can be created in a laboratory setting.
- All of the Flerovium isotope are unstable and radioactive.
- All isotopes of Flerovium decay via alpha decay or spontaneous fission, but none of them undergo beta decay.
- The atomic mass of Flerovium isotopes are: 284Fl, 285Fl, 286Fl, 287Fl, 288Fl, 289Fl, and 290Fl.
- Isotopes are created when a heavier element decays or when two light nuclei fuse together.
- The heavier isotopes are more stable than the lighter ones.
- Most Flerovium isotope decay by emitting alpha particles into Copernicium isotopes.
- The most stable isotope, 289Fl, has a half-life of 2.6 seconds.
- Flerovium-298, a flerovium isotope with a longer half-life, is predicted but has yet to be discovered.
Physical Properties of Flerovium
- Flerovium’s instability makes it difficult to conduct an objective examination of its physical properties.
- Given its swift disintegration, only a few properties of Flerovium have been studied to date.
- Flerovium is a synthetic, super-heavy transactinide element.
- It is found in the 7th period, the 14th Group, and the p-block of the periodic table.
- Flerovium is expected to be found in the carbon group (Group 14), among silicon, tin, germanium, and lead. Its characteristics are expected to be those of the carbon group elements.
- The melting point and the boiling point of the element 114 is yet to be known.
- The atomic mass of Flerovium is anticipated to be 289. (The atomic mass of man-made trans-uranium elements is calculated using the periodic table’s longest-lived isotope. These atomic weights should be considered tentative because a new isotope with a longer half-life may be created in the future).
- Flerovium was expected to be a gas under normal conditions, similar to copernicium, however recent study has claimed that it is a noble metal.
- Flerovium is projected to crystallize into a face-centered cubic shape.
Chemical Properties of Flerovium
- Flerovium is a highly radioactive element. Its chemical properties have yet to be completely investigated. Isotopes have short half-lives, and the compounds they contain are highly volatile, making conclusive chemical analysis challenging.
- Flerovium is currently the heaviest element that has had its chemistry studied experimentally yet nothing is conclusive.
- Early atomic computations on element 114 (flerovium, Fl), with closed and quasi-closed electron shell configurations of and 6d107s27p2, anticipated that it would be noble-gas-like due to very strong relativistic effects on the 7s and 7p2 valence orbitals.
- Recent fully relativistic computations of Cn and Fl in various settings reveal that they are less reactive than their lighter homologues in the groups, but retain a metallic quality.
- Experimental gas-solid chromatography research on Fl deduced the establishment of a weak link upon physisorption on an Au surface from the earliest trials.
- Fl is projected to be a noble metal. (The chemical elements that exist in solid metal form, are exceptionally resistant to oxidation and high temperatures, have anti-corrosive properties, and do not react strongly with acids.)
- Flerovium is projected to have the highest initial ionization energy of any element in its group. Furthermore, it would have a stronger electronegativity than lead.
- Other members of the carbon group create stable compounds with a +4 oxidation state, however flerovium is projected to be most stable at +2 or 0.
- The observed adsorption behavior of Fl indicates a stronger inertness than its nearest homologue in the group, Pb.
- However, the reported lower limit for the adsorption enthalpy of Fl on an Au surface indicates the establishment of a metal-metal link between Fl and Au.
- Fl is the least reactive element in the group, although it remains a metal.
Synthesis of Flerovium
- All elements with atomic numbers more than 100 can only be created through reactions in a particle accelerator, such as a cyclotron; they do not develop in a nuclear reactor.
- Flerovium-290 is produced by bombarding plutonium-244 with calcium-48.
Uses of Flerovium
- Given that few atoms of this metal have been synthesized thus far, there are currently no specific or specialized applications for Flerovium other than scientific research.
- A constant experimental study aimed at achieving an obvious conclusion requires a large number of atoms. Perhaps a few Flerovium atoms have been produced thus far.
Health Effects of Flerovium
- Flerovium is a very unstable chemical; when created, it swiftly decomposes into other elements, therefore it has no impact on human health. However, being a radioactive element it must be toxic.
Environmental Effects of Flerovium
- Flerovium’s environmental effects are negligible due to its short half-life (just a few seconds).
Video Reference
References
- https://www.theguardian.com/science/grrlscientist/2013/nov/29/grrlscientist-elements-flerovium-114
- https://pubs.acs.org/doi/10.1021/ic4026766
- https://periodic-table.com/flerovium/
- https://www.chemicool.com/elements/flerovium.html#google_vignette
