Ion exchange chromatography (IEC), along with ion-partition/interaction and ion-exclusion chromatography, is an important analytical technique for the separation and determination of ionic compounds. This separation is based on ionic (or electrostatic) interactions between ionic and polar analytes, ions in the eluent, and ionic functional groups fixed to the chromatographic support. Ion exchange due to competitive ionic binding (attraction) and ion exclusion due to repulsion between similarly charged analyte ions and the ions fixed on the chromatographic support both play a role in ion chromatography separation. Because of their varying charge properties, biomolecules interact differently with charged chromatography media.
What is Ion Exchange Chromatography?
Ion exchange chromatography (also known as ion chromatography) is a method of separating ions and polar molecules based on their affinity for ion exchangers.
The reversible exchange of ions between the target ions present in the sample solution and the ions present on ion exchangers is thus the separation principle.
Anionic and cationic exchangers, two different types of exchangers, may be used in this process.
Cationic exchangers have positively charged groups that attract positively charged cations. Because their negative charges result from the ionization of an acidic group, these exchangers are also known as “Acidic ion exchange” materials.
Anionic exchangers have positively charged groups that attract negatively charged anions. These materials are also known as “basic ion exchange” materials.
Types of Ion Exchange Chromatography
Cation-exchange chromatography: It separates molecules according to their net surface charge. It employs a negatively charged ion-exchange resin that is attracted to molecules with net positive surface charges.
Anion-Exchange Chromatography: It is a technique for separating molecules based on their net surface charge. It employs a positively charged ion exchange resin that is attracted to molecules with net negative surface charges.
Principle of Ion Exchange Chromatography
Ion-exchange chromatography is used to separate charged biomolecules. The charged analyte samples will be used as a liquid phase. When the analyte passes through the chromatography column in the stationary phase, it is opposite the charged sites. The analytes that have been isolated based on their charge are eluted using a solution of varying ionic strength. The analytes are very selectively separated according to their different charges by moving such a mobile phase through the chromatography column.
How does Ion exchange chromatography work?
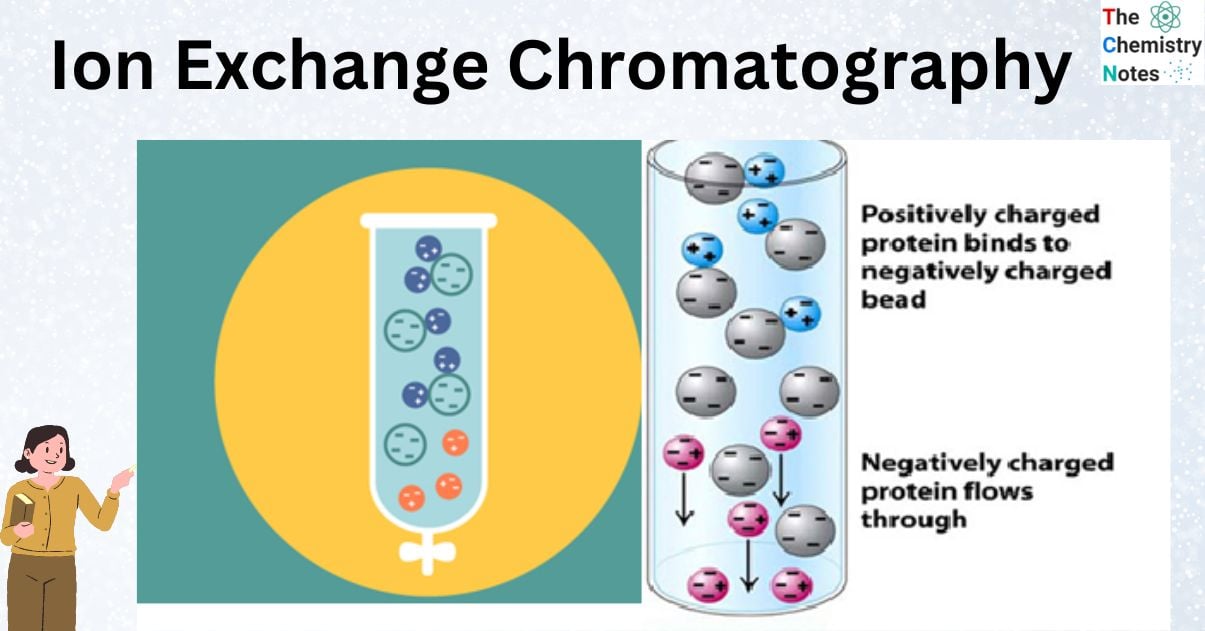
Functional groups in biomolecules can ionize in solution and give the molecule a particular net charge. Proteins, for example, are composed of amino acids with both amino (-NH2) and carboxylic acid (-COOH) functional groups. A protein’s 3D structure determines which amino acid residues are exposed on its surface. These residues ionize depending on the pH of the medium, giving the molecule positive and negative surface charges. More amino groups are protonated at low pH levels, and protein molecules have a net positive charge. At higher pH values, however, more carboxylic acid groups are deprotonated, and the resulting anions negatively charge the surface of the protein molecules.
The net surface charge is determined by the total number of ionized functional groups present on the surface, and each molecule has a unique net surface charge at any given pH value. At its isoelectric point (pI), which is a specific pH value at which it has no net charge on the molecule, a protein is electrically neutral.
[Image Source: Analysis and Separation]
The oppositely charged ions that are present on the surface of the stationary phase particles are electrostatically bound to the polar or charged analytes when they are loaded into an ion-exchange column. The electrostatic attraction to the oppositely charged particles of the stationary phase will be stronger the more positive or negative charges are on the surface of the analyte molecules. The target analyte retention time is also influenced by how many times it interacts with the stationary phase. By adjusting the pH or ionic strength, aqueous mobile phases with buffers and salts are used to elute the bound analytes.
Procedure of Ion Exchange Chromatography
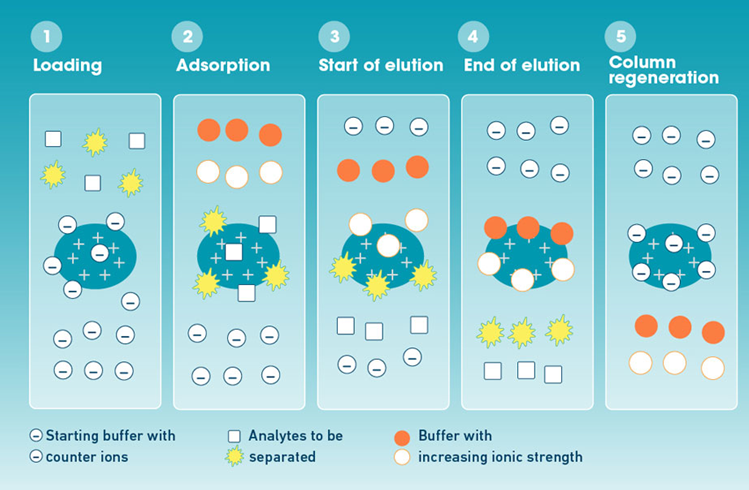
Equilibration
The stationary phase is first washed with the start buffer (original buffer composition) until the baseline is stabilized and the eluent pH is constant. By doing this, you can be sure that the ionizable groups on the column are open to interacting with the molecules of the charged analyte.
Sample loading
Samples are injected into the column after being dissolved in start buffer or buffers with a pH similar to the start buffers. The buffer’s pH and ionic strength are adjusted so that only the analytes bind to the column and not impurities.
Wash
The column is once more washed with the initial buffer to get rid of any molecules that aren’t charged or have the same charge as the stationary phase. Following the impurities, the baseline stabilizes.
Elution
The bound analytes are released from the column surface using a salt gradient as the ions in the elution buffer compete with and replace the analytes on the charged sites. Weakly bound analytes (molecules with lower surface charge densities) begin to elute from the column at low ionic strengths. Strongly bound molecules with increasing surface charge densities sequentially elute from the column as the salt concentration is raised. The bound analytes can also be released from the column at their respective pI value by using a pH gradient. Cations are eluted from the anion exchange column by decreasing the pH of the eluting buffer, whereas anions are eluted from the column by increasing the pH of the eluting buffer. a gradient in pH.
Column regeneration
By washing off any molecules that have become stuck to the column, the column capacity is finally recovered for the following run. This is accomplished by allowing a buffer with a high ionic strength to pass through the column up until the baseline and pH of the eluent stabilize. Prior to the subsequent run, the column is then conditionally set with the starting buffer.
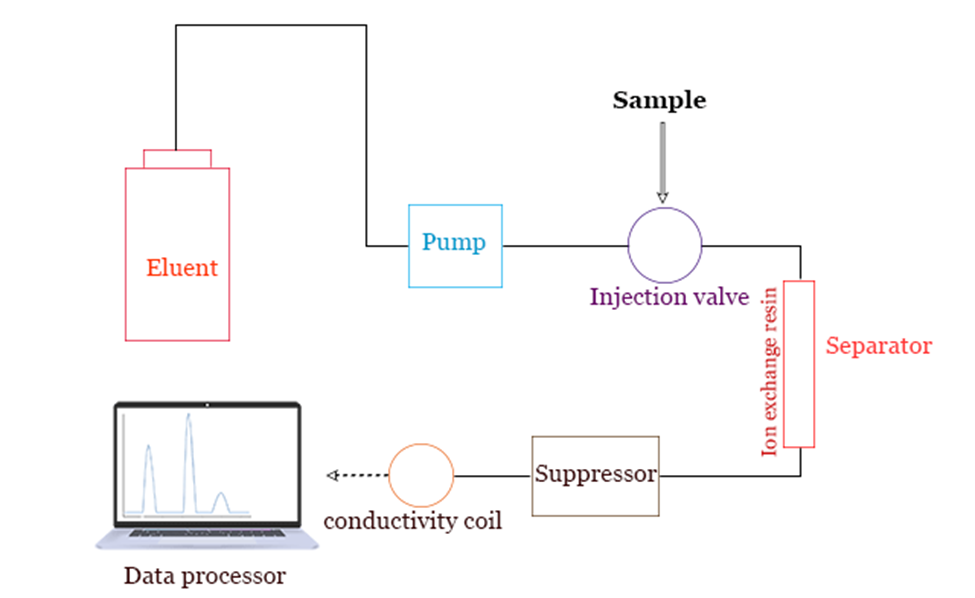
Instrumentation of Ion Exchange Chromatography
[Image Source: Learning Chemistry]
Ion exchange column chromatography typically includes:
Pump: A high-pressure pump is one of the essential components of an ion exchange chromatography system because it maintains a steady flow of eluent.
Injector: The easiest method of introducing samples into the eluent stream and onto the column is through the use of an injection valve, or injector. A liquid sample should be able to be injected with high reproducibility and pressure using injectors with a volume range of 0.1 to 100 ml (up to the 4000 psi).
Columns: For the separation of the sample mixture, a column is chosen based on the area of application. The columns used in laboratories are made of glass, while the columns used in industries are either made of premium stainless steel or polymer.
Detector: This determines the analyte peaks in the column eluent.
Factors Affecting the Separation in Ion Exchange Chromatography
Column: To achieve effective separation, the right column must be chosen because the porosity and size of the polymeric resin affect the resolution. While offering better resolution, smaller particle sizes also result in more system backpressure. In the early stages of protein purification, for instance, columns stuffed with large-sized beads are used, while smaller beads and slower flow rates are used for the final purification.
Buffers: Typical buffers used as mobile phases for cation exchange chromatography include formate (3.8), acetate (4.6), MES (6.1), phosphate (7.2) or tris (8.1), while buffers that are used as mobile phases for anion exchange chromatography include tris, piperazine (9.7) and diethylamine (11). When choosing buffers for IEC, the pka values should be taken into consideration.
Ionic strength/salt concentration: Elution is typically performed in buffers containing 300–500 mM of salts, such as sodium chloride, potassium chloride, sodium bromide, sodium sulfate, or sodium acetate.
Additives: When necessary, additives like urea, sugars, or detergents are added to make the analytes more soluble, stop them from precipitating, or ensure analyte stability by inhibiting enzyme activity. However, at the operating pH, the additives may become charged and may obstruct the separation.
Sample load: As advised by the manufacturer, the mass of the analyte(s) should not be greater than the column’s binding capacity in order to achieve optimal resolution.
Gradient: To obtain the desired resolution, the run time and percentage change of ionic strength over time must be optimized. Since a linear pH gradient is challenging to accurately and consistently create, step pH gradients are frequently preferred.
Flow rate: To increase throughput, use the highest flow rate at which the resolution is unaffected. In order to speed up column regeneration and re-equilibration, higher flow rates can be used.
Ion Exchange Chromatography Applications
The most popular chromatographic technique for cation and anion separation and purification is ion exchange chromatography. It is primarily used for the purification or separation of proteins, amino acids, water softening, and metal separation.
- It is employed in a variety of industries, including the pharmaceutical, petrochemical, and agricultural sectors, in addition to analytical chemistry and biological chemistry for the separation of cations and anions.
- It is employed in medicine to analyze sugar and amino acids. The division and purification of blood components is another use for it.
- In the chemical industry, it is used for quality control or impurity analysis.
- It is used in agriculture to analyze the micronutrients in soil samples.
- It is used to identify sulfur compounds in the petrochemical sector.
- Used in environmental chemistry for the identification and separation of toxic pollutants found in aquatic and atmospheric environments.
Advantages of Ion Exchange Chromatography
- This method of separating charged particles is the most effective.
- This is applicable to almost any charged molecule, including nucleotides, small amino acids, and large proteins.
- It is carried out in the lab for both analytical and preparatory purposes.
- This method can also be used to isolate inorganic ions.
- It is a practical and effective method for water softening.
Disadvantages of Ion-Exchange Chromatography
- The need for a buffer is one of the main drawbacks of ion-exchange chromatography.
- By using this technique, only charged molecules can be separated.
- For the separation of components, it needs a buffer.
- Its operating expenses are high.
- Sodium ions entering the softened water can increase its acidity level.
- To maintain capacity and resolution, weak ion exchange columns must be used within the recommended pH range because they are sensitive to changes in pH.
- High salt concentrations may necessitate the use of bio-inert LC systems to prevent corrosion.
- Due to the high salt concentration in the eluent, coupling with mass spectrometry (MS) is not as effective.
References
- Tatjana Weiss; Weiss, Joachim (2005). Handbook of Ion Chromatography. Weinheim: Wiley-VCH. ISBN 978-3-527-28701-7.
- Ion-Exchange Chromatography: Basic Principles and Application. https://doi.org/10.1007/978-1-4939-6412-3_11
- Gjerde, Douglas T.; Fritz, James S. (2000). Ion Chromatography. Weinheim: Wiley-VCH. ISBN 978-3-527-29914-0.
- Jackson, Peter; Haddad, Paul R. (1990). Ion chromatography: principles and applications. Amsterdam: Elsevier. ISBN 978-0-444-88232-5.
- https://www.technologynetworks.com/analysis/articles/exploring-the-principle-of-ion-exchange-chromatography-and-its-applications-357491
- https://whatishplc.com/hplc-basics/principle-and-procedure-of-ion-exchange-chromatography/
- Acikara ÖB. Ion-Exchange Chromatography and Its Applications. Chapter 2. Column Chromatography, Dean F. Martin and Barbara B. Martin. IntechOpen. 2013. doi:10.5772/55744
