Peptides are short chains of amino acids that typically range from 2 to 50 amino acids in length. They are protein variants that are shorter in length. The names of the constituent amino acids are written from N-terminal to C-terminal during the peptide naming process. N-terminal amino acids have free amino groups, while C-terminal amino acids have free carboxyl groups.
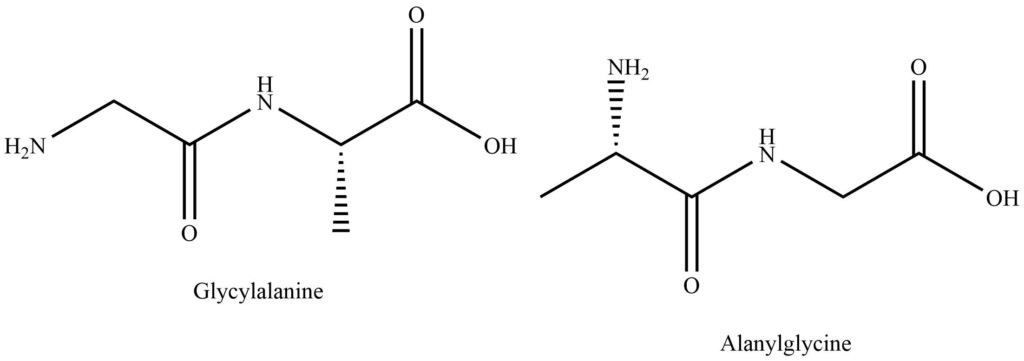
The suffix ‘ine’ is replaced by -Yl in the names of all amino acids except C-terminal acid. Polypeptide names are sometimes abbreviated using three letters to represent constituent amino acids.
Structure of peptides
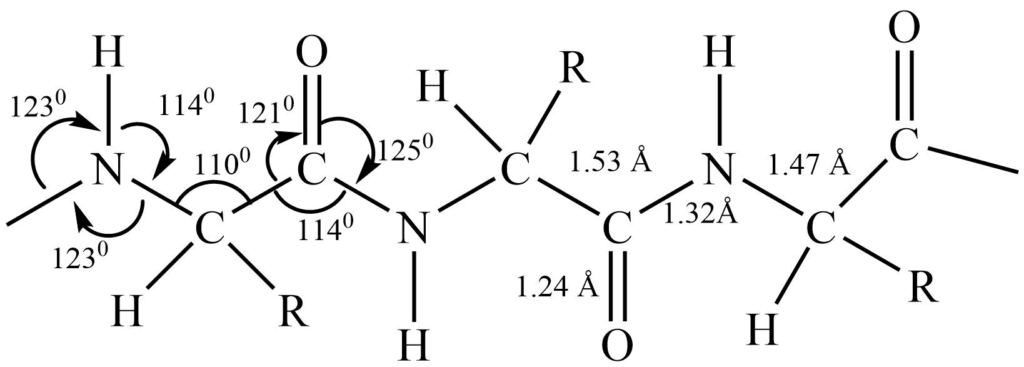
X-ray diffraction analysis of amino acids shows that peptide linkage is flat that is carbonyl carbon, nitrogen, and all atom attached to them are in the same plane. The bond length between carbon and nitrogen i.e. C-N bond length comes out to be 1.32 A, apart from the usual C-N single bond length of 1.47 A. This shows that the nitrogen bond has a 50% double bond character. Bond angle measurement shows that the bonds to nitrogen are similar to those of trigonal carbon atoms.
Types of peptides
on the basis of production
- Milk peptides: Peptides obtained from the breakdown of milk protein
- Peptones: these are obtained from the proteolysis of animal milk and meat. Sometimes also obtained from vitamins and fat.
- Ribosomal peptides: These peptides are formed during the formation of mRNA by the translation process. E.g., Intestinal peptides, Pancreatic peptides etc.
- Non-ribosomal peptides: Non-ribosomal peptides are made up of enzymes that are unique to each peptide rather than the ribosome.
- Peptide fragments: Peptide fragments are protein fragments used to identify the source of protein.
on the basis of the number of amino acids present
- Dipeptides: Dipeptides are the peptides formed by the linkage of two amino acids by peptide linkage.
- Tripeptides: Tripeptides peptides are the peptides formed by the linkage of three amino acids by peptide linkage.
- Tetrapeptides: Tetrapeptides Consist of four amino acids linked by a peptide linkage.
- Oligopeptides: Oligopeptides contain amino acids not more than ten in their chain.
- Polypeptides: Polypeptides consist of more than amino acid residues up to 100.
- Macropeptides: Macropeptides consist of amino acid residues of more than 100.
Determination of the structure of peptide by terminal amino acid residue analysis
The amino acid residues present at the two ends of the peptide chain are different from all amino acids and also from each other. The amino acid present at one end containing the free -NH2 group is called N-terminal amino acid. While the amino acid present at another end with free -COOH is called a C- terminal amino acid.
Two structures differ in the respect that the first N-terminal amino acid is glycine and the C-terminal amino acid is alanine. While in the second peptide N-terminal amino acid is alanine and the C-terminal amino acid is glycine. Various methods have been developed to remove the terminal amino acid residue from the peptide chain and to establish the arrangement of various amino acids in the peptide chain.
N-terminal residue analysis:
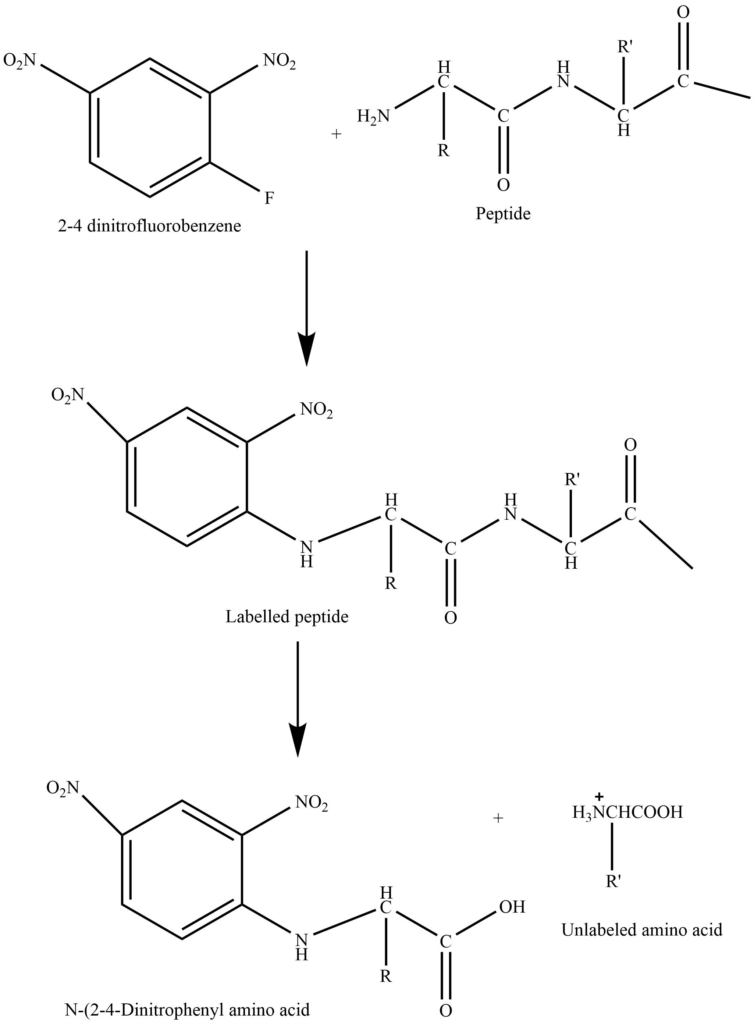
1. Sangar method: Sangar first used 1-fluoro-2-4-dinitrobenzene(FDNB) for the determination of amino acids constituted the amino end of the polypeptide.
In this method, the polypeptide is treated with a sangar reagent at room temperature in the presence of sodium hydrogen carbonate solution to produce the polypeptide’s 2,4 -dinitrophenyl derivative. The resulting derivative is hydrolyzed by acid to yield the dinitrophenyl derivative of the N-terminal amino acid as well as the remaining amino acid residues. The dinitrophenyl derivative of amino acid is isolated and analyzed chromatographically.
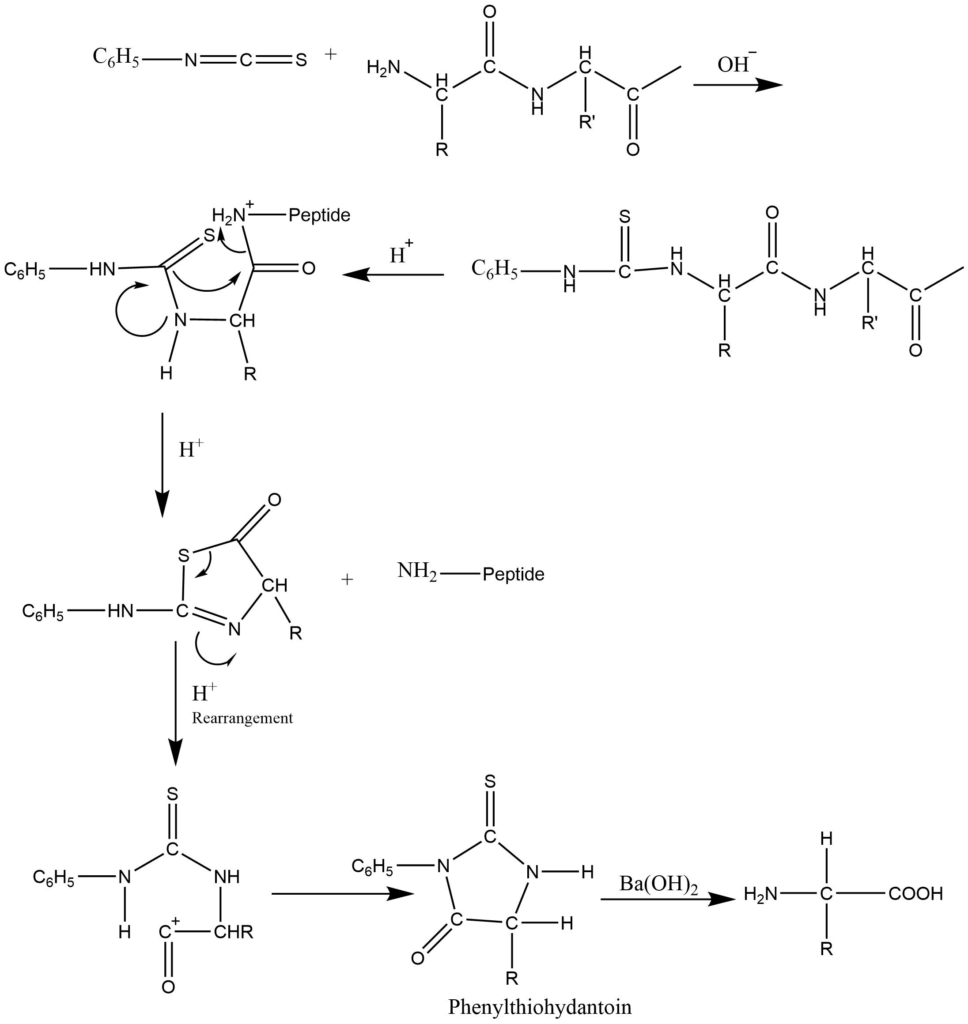
2. Edman’s method: The polypeptides are treated with Edman’s reagent i.e., phenylisothiocyanate in the presence of dilute alkali to form N-phenylthiocarbomyl peptide, which rection with mild acid, N-terminal amino acid is removed as phenylthiohydantoin while Other amino acid residue remains attached. The phenylthiohydantoin is separated from the reaction mixture and compared with phenylthiohydantoin obtained from the known amino acids.
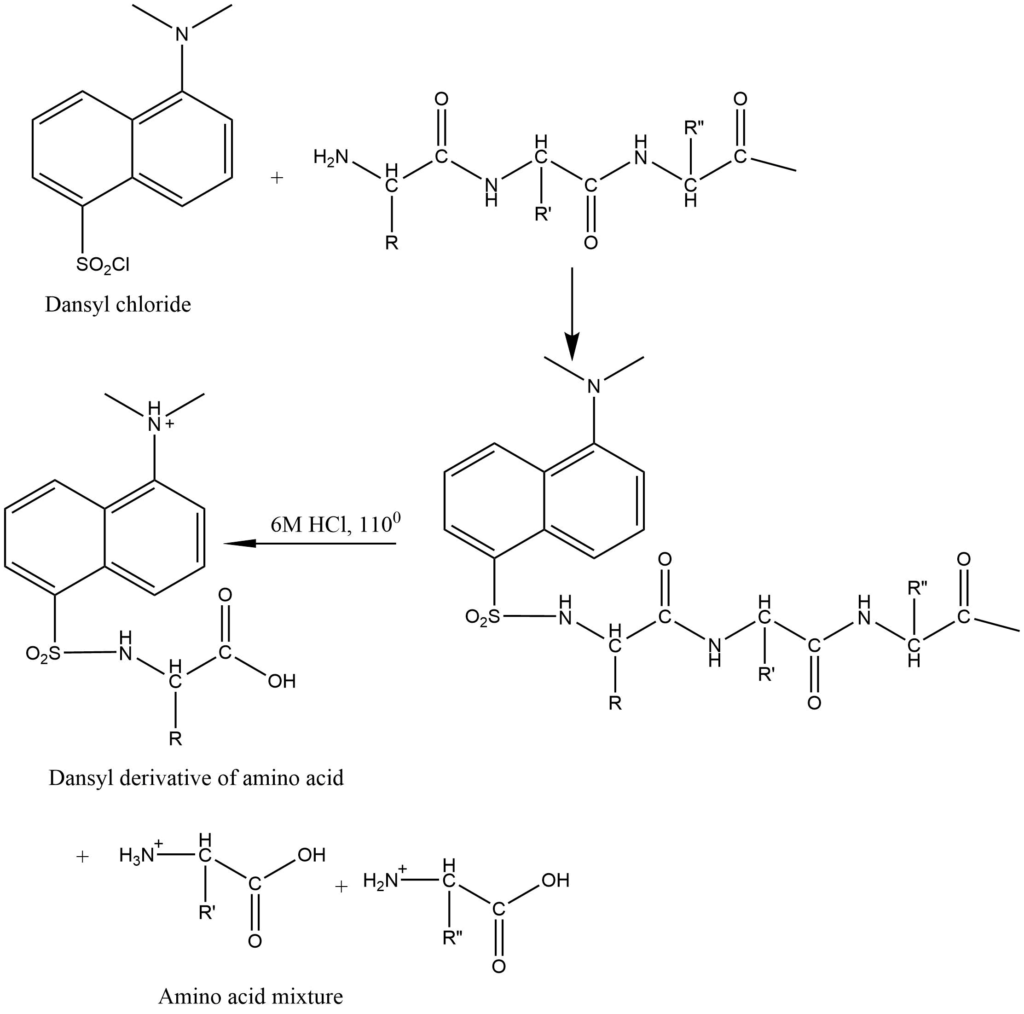
3. Dansyl method: Here 1-dimethylamino naphthalene-5-sulphonyl chloride i.e. Dansyl chloride is used for the determination of N-terminal amino acid residue.
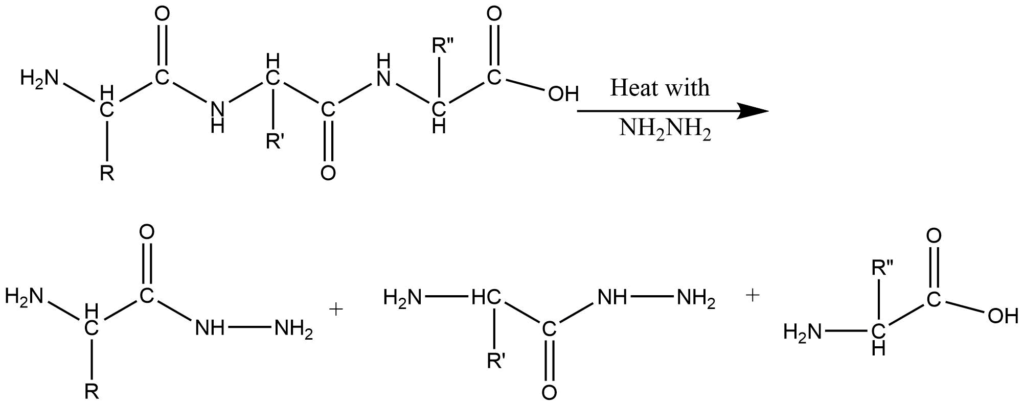
C-terminal residue analysis:
1. Hydrazinolysis method: In this process, the peptide is heated with anhydrous hydrazine at the temperature of 373K. The amino acid residues except for C-terminal, are converted to hydrazides while the C-terminal residue is obtained as a free amino acid.
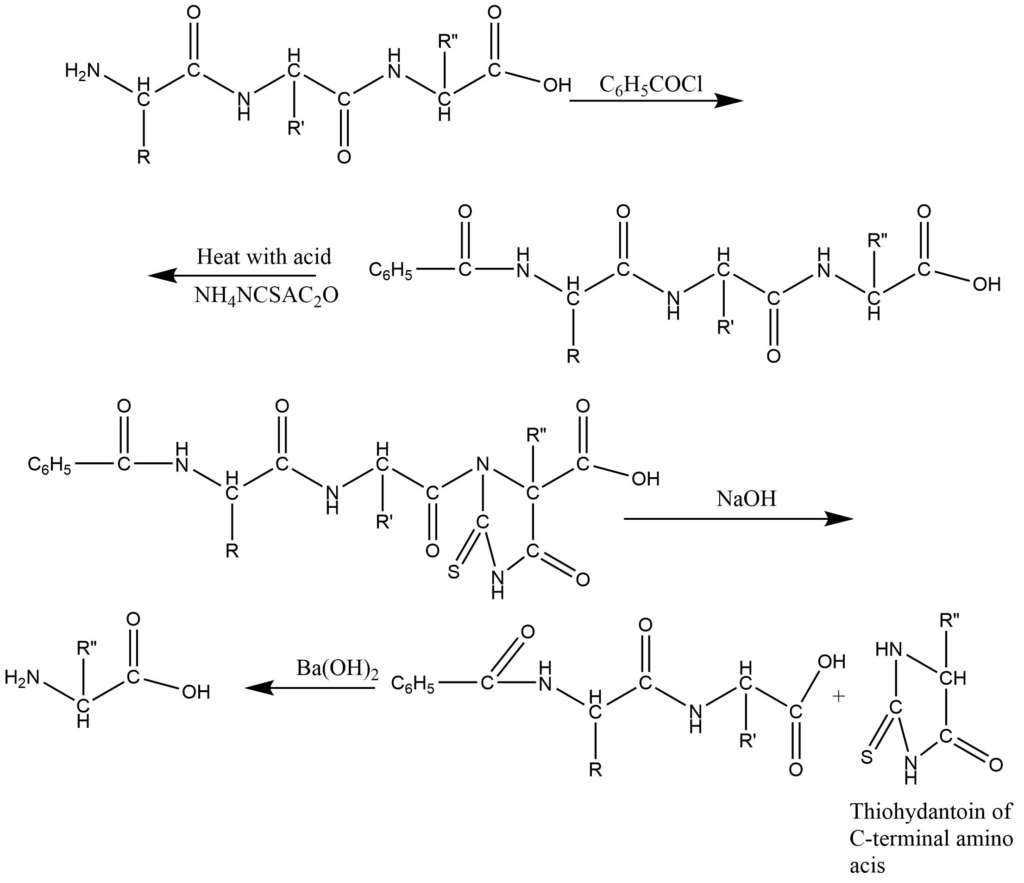
2. Schlack and Kumpf method: In this process, the amino acid group of N-terminal residue is protected by benzoylation and then the C-terminal residue is converted into thiohydantoin and hydrolyzed to amino acids.
3. Reductive method: reaction of a peptide with lithium aluminum hydride or lithium borohydride reduces the free terminal carboxyl group into the primary alcoholic group. Which on further hydrolysis gives amino alcohol from C-terminal amino acid along with the mixture of amino acids. The amino alcohol is separated and identified by using paper chromatography.
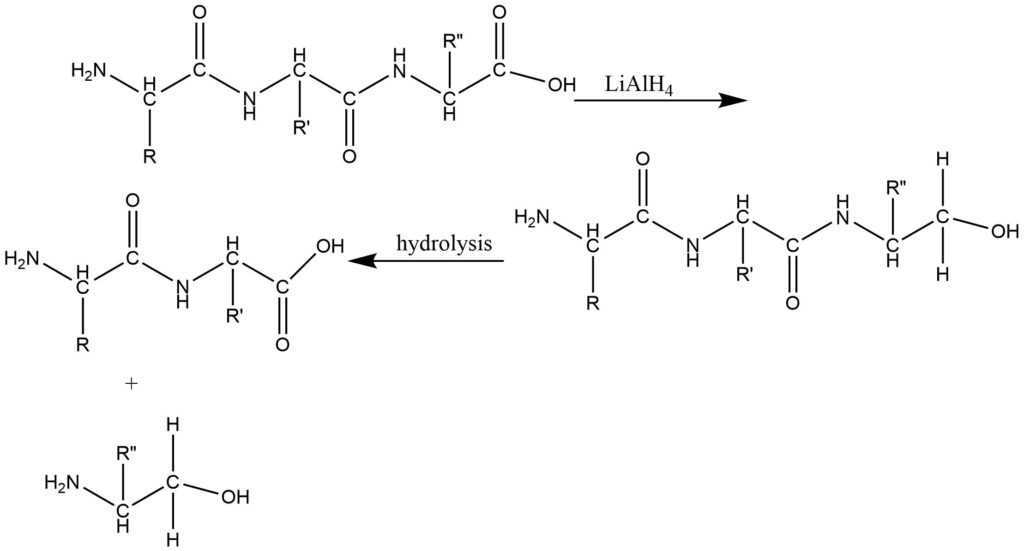
Synthesis of Peptides
Peptides having higher molecular weight are generally prepared by polymerization of dipeptides and tripeptides. The synthesis of peptides from the amino acid involves different steps they are:
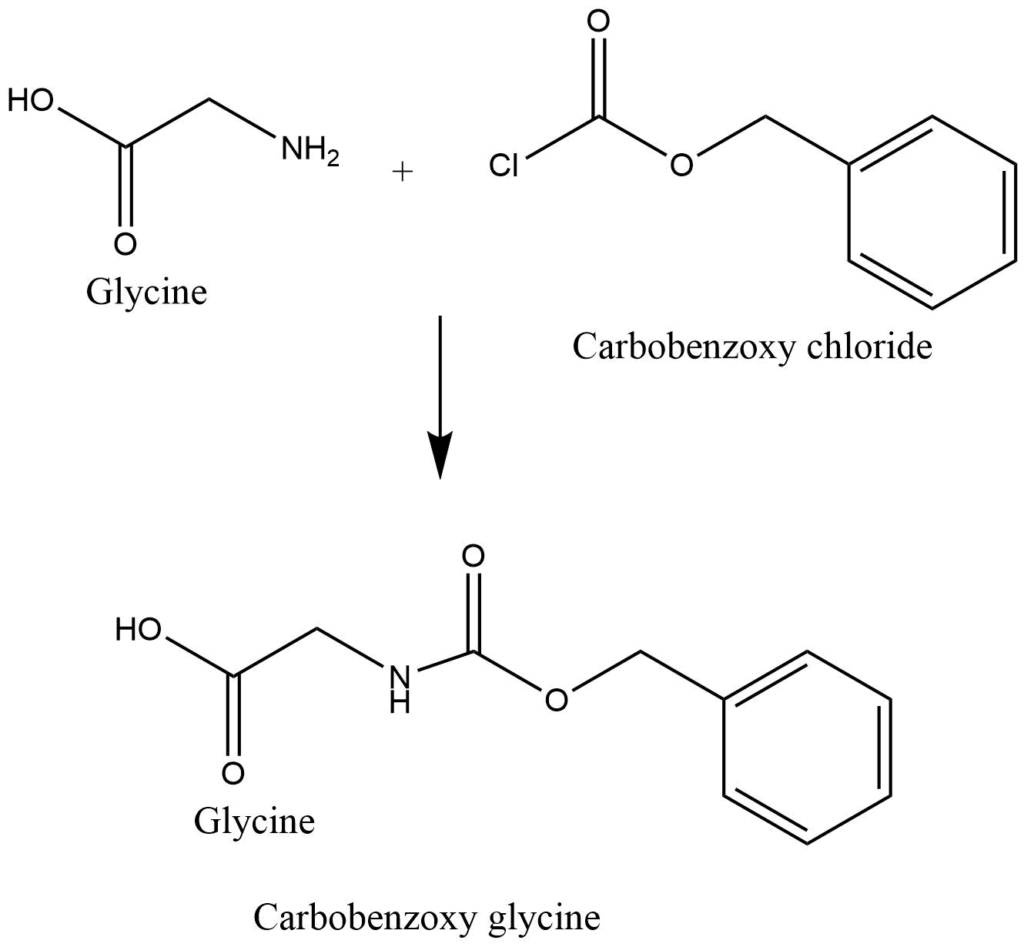
1. Protection of amino i.e. – NH2 group of amino acid with carbobenzoxy chloride.
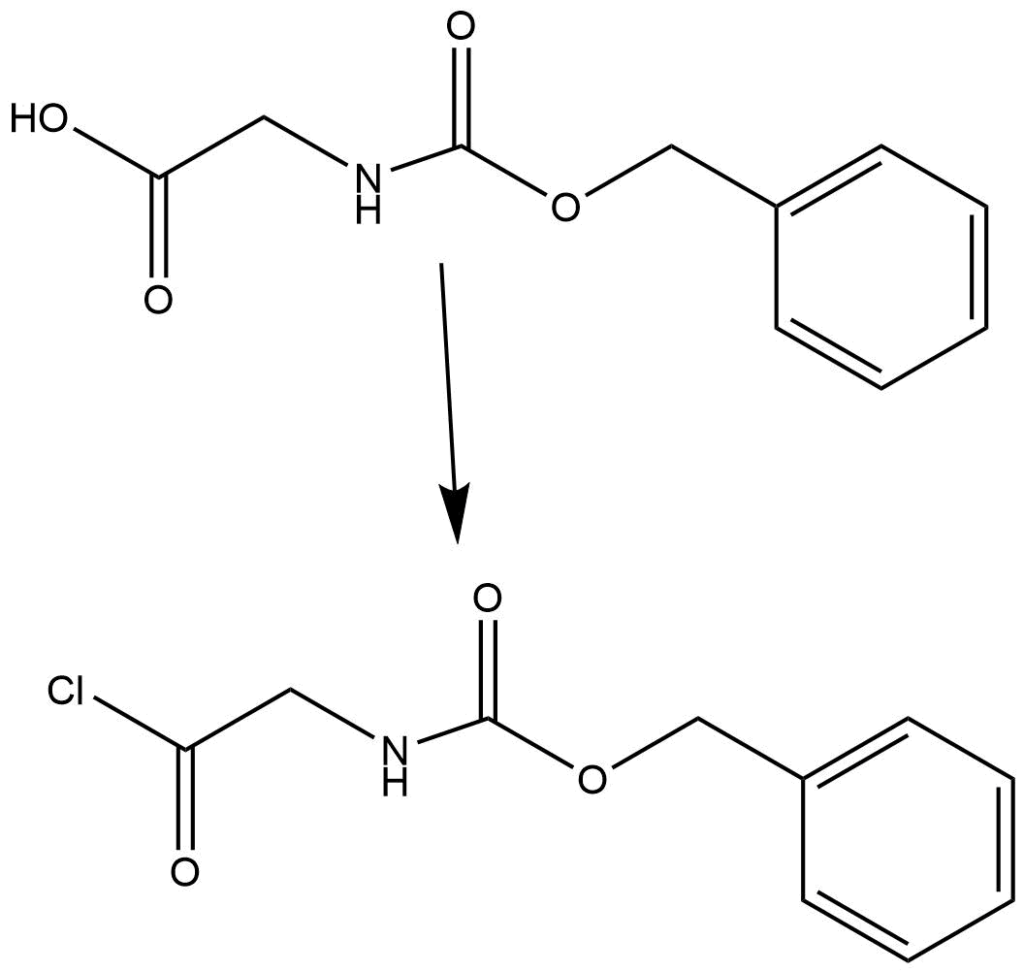
2. Conversion of a carboxyl group into acid chloride by reacting with thionyl chloride (SOCl2).
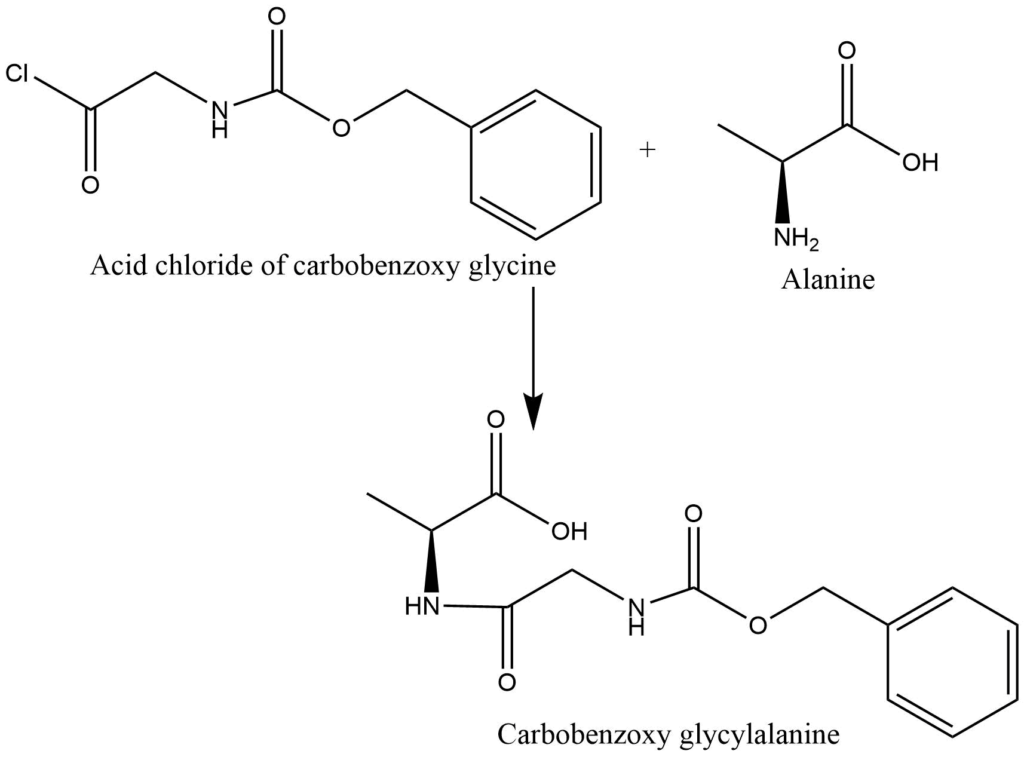
3. formation of peptide linkage
4. removal of chlorobenzoxy group
Peptides supplement
The peptides can be obtained from plant and animal sources of protein. These are
- Egg
- Oats
- Milk
- Wheat
- Soy
- Fish
- Meat etc.
Some common peptide supplements are:
- Collagen peptides: Benefits the skin health and act as anti-aging.
- Creatine peptides: Build strength and muscle mass.
Benefits of peptides
- Peptides act as precursors of proteins.
- They act as alkaloids, hormones, antioxidants as well as growth factors.
- Increase skin firmness.
- Peptides protect skin from UV damage.
- Growth hormone realizing hormonal peptides show antioxidant activity and are used for the treatment of alcohol addiction, sexual dysfunction, and mood disorders.
- Peptides can help to cure inflammation and repair skin damage.
- Some peptides are antimicrobial that kill the bacteria causing acne.
References
- https://www.onlinebiologynotes.com/peptide-types-functions/
- https://www.vedantu.com/chemistry/peptides
- https://byjus.com/chemistry/peptides/
- https://byjus.com/jee/peptide-bond/
- https://conductscience.com/classes-and-benefits-of-peptides/
- https://www.healthline.com/health/peptides-for-skin
- https://www.aspirerejuvenation.com/blog/benefits-of-peptides-hgh/
- https://www.medicalnewstoday.com/articles/326701#side-effects
