Visually tracking physical changes and chemical reactions can be done well with chemical reaction equations. However, we must first understand how to properly do representation of chemical reactions before we can evaluate these equations to see these changes. To put it another way, we must develop our ability to balance equations. Our conclusions will be flawed if an equation is unbalanced. How can we simply learn to balance equations? For the representation of chemical reactions we need to learn about the symbols, molecular formulas, balancing equations, charges balance, and many more.

Chemical Equations
The symbolic representation of a chemical reaction is a chemical equation. For the same, the reactants’ and products’ formulas are represented by symbols. For instance, the following is how the combustion of methane gas might be expressed in words:
The word “equation” refers to this formula. You can convert the word equation into a chemical equation by writing the substance’s symbol and formula in instead of its name.
CH4 (g) + 2O2 (g) + Heat ⟶ CO2 (g) + 2H2O (l)
(Methane) (Oxygen) (Carbon dioxide) (Water)
You can better comprehend chemical processes by using the symbols for the elements.
- Carbon, hydrogen, and oxygen are all used in this process.
- Methane and oxygen are represented chemically in this reaction by the letters CH4 and O2, respectively.
- Carbon dioxide and water are the results of this process.
Symbolic Representation of Chemical Reactions
| Symbol | Meaning |
| + | used to differentiate between different reactants or products |
 | when reading the equation, it is pronounced “yields” or “produces” to distinguish the reactants from the products. |
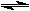 | Equilibrium arrow, which is employed when the reaction can run in both directions, will be used later in the course. |
| (g) | substance is in a gaseous state |
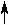 | a different method of describing a substance in its gaseous state |
| (s) | substance is in a solid state |
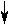 | a different method of describing a substance is in its solid state |
| (aq) | indicates that the substance is dissolved in water – the aq comes from aqueous |
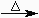 | indicates that heat is applied to make the reaction proceed |
Arrow in Chemical Reactions
An energy requirement for a reaction is typically displayed above the arrow. The reactants and products of a chemical equation are located on the left and right sides, respectively. In cases where the reaction is bidirectional, it is appropriate to indicate this by depicting arrows pointing in both directions. In general, it is advisable to rephrase the equation in cases where the direction of the arrow is from right to left.
A capital Greek letter delta (Δ) is inscribed over the reaction arrow if energy is delivered to the reaction in the form of heat; hv is inscribed over the reaction arrow if energy is supplied in the form of light.
Types of Arrow in Chemical Reactions
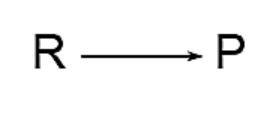
Right Arrow: The rightward pointing arrow is the predominant symbol utilized in chemical reaction equations. The orientation denotes the direction of the reaction.
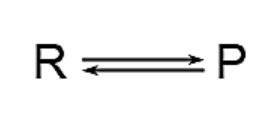
Double Arrow: The presence of a double arrow in a chemical equation indicates a reversible reaction.
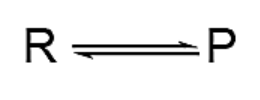
Equilibrium Arrow: When the reaction is at balance, two arrows with a single spike pointing in opposite directions show that the reaction can be stopped and started again.
Balancing Atoms
To balance chemical equations, we use the rules of conservation of atoms and electrons. A chemical equation is said to be balanced if the amount of each element and the amount of electrical charge in the reactants and the products are the same. Here are the steps to do balance the atoms:
- If the number of atoms on each side is the same, then everything is fine and the equation is balanced.
- If the number of atoms on each side is not the same, you need to use big (whole) numbers in front of each element or combination to make them equal.
- When you add a large number, you multiply the whole combination by that number, so you have to change the numbers in the table to reflect that.
- Repeat until the number of atoms on the left is the same as the number on the right.
Balancing Chemical Equations
In a balanced chemistry equation, the number of atoms on both sides is the same. Here’s an example, zinc forms zinc sulfate and hydrogen when it combines with diluted sulfuric acid.
Zn + H2SO4 → ZnSO4 + H2
Both the reactants and the products have zinc in them. Both the products and the reactants have two hydrogen atoms, and both have only one sulfur atom. Last, there are four oxygen atoms in both the reactants and the result. In the above molecular equation, this means that there are the same number of atoms in both the reactants and the products.
Balancing Charge
When balancing numbers, it is also important to think about the law of conservation of electrons.
When it comes to ionic substances, balancing charge is the most important thing to do. You might remember that ionic molecules are made up of both an anion and a cation. When an ionic compound comes into contact with water (when an aqueous solution is made), the compound likes to ionize quickly. This means that the ionic compound breaks apart into its individual ions. Ionization is the name for this process.
Let’s look at what happens when we put table salt in water as an example.
NaCl → Na+ + Cl–
As you can see, salt breaks down into a sodium ion and a chloride ion.
When we work with ionization formulae, we have to balance both the atoms and their charges:
| amount in products | amount in reactants | |
| sodium | 1 atom | 1 atom |
| chlorine | 1 atom | 1 atom |
| charge | 0 | +1 + -1 = 0 |
How to Balance Chemical Equations
To balance a chemistry equation, you only need to do four simple things:
- Write out the equation that isn’t balanced to show the reactants and the products.
- Write down the number of atoms of each element on each side of the arrow that shows the process.
- Add the factors (the numbers in front of the formulas) so that on both sides of the equation, the number of atoms of each element is the same. The hydrogen and oxygen atoms are the ones that are easier to balance.
- Check your work by writing down the states of the reactants and the products.
- Write an Unbalanced Chemical Equation.
The unbalanced chemical equation must first be written down. If you’re asked to balance a chemical equation but simply given the names of the products and reactants, you’ll need to search them up or use compound naming conventions to figure out their formulas.
Let’s experiment with a real-life reaction: iron rusting in the air. You must first identify the reactants (iron and oxygen) and the products (rust) in order to write the reaction. Write the unbalanced chemical equation:
Fe + O2 → Fe2O3
Take note that the reactants are always on the left side of the arrow. They are separated by a “plus” sign. Following that is an arrow denoting the reaction’s direction (reactants become products).
- Write the number of atoms.
The next stage in balancing the chemical equation is to count the number of atoms of each element on each side of the arrow:
Fe + O2 → Fe2O3
Remember that a subscript specifies the number of atoms. For example, O2 contains two oxygen atoms. Fe2O3 contains two iron atoms and three oxygen atoms. Fe contains one atom. When there is no subscript, there is only one atom.
On the reactive end: 1 Fe and 2 O
On the product front: 2 Fe and 3 O
Because mass is not created or destroyed in a chemical reaction, you must add coefficients to the chemical formulas to alter the amount of atoms so they are the same on both sides.
- Adding Coefficients to Balance Mass in a Chemical Equation
Subscripts should never be changed while balancing equations. You multiply the coefficients. Coefficients are multipliers of whole numbers. If you write 2 H2O, for example, you have double the number of atoms in each water molecule, which is 4 hydrogen atoms and 2 oxygen atoms. As with subscripts, the coefficient of “1” is not written, therefore if you don’t see one, it signifies there is just one molecule.
There is an approach that can help you balance equations faster. It is known as balance by examination. Essentially, you take the number of atoms on each side of the equation and add coefficients to the molecules to balance out the amount of atoms.
First, balance the atoms in a single molecule of reactant and product.
Last, balance any oxygen or hydrogen atoms.
Fe + O2 → Fe2O3
Because iron is present in both the reactant and the product, its atoms must be balanced first. Because there is one iron atom on the left and two on the right, you might suppose that adding 2 Fe on the left would work. While this would balance iron, you already know you’ll need to alter oxygen as well because it’s not balanced. You know you have to discard a coefficient of 2 for some higher number just by looking at it.
4 Fe + O2 → 2 Fe2O3
With four iron atoms on each side of the equation, iron is balanced. The next step is to balance the oxygen. Last, balance the oxygen and hydrogen atoms.
The final stage in balancing chemical equations is to add coefficients to oxygen and hydrogen atoms. The reason for this is that they frequently appear in many reactants and products, thus tackling them first usually results in extra labor.
Examine the equation (using inspection) to determine which coefficient will work to balance oxygen. If you substitute a 2 for O2, you will get 4 atoms of oxygen, but the result has 6 atoms of oxygen (coefficient of 2 multiplied by subscript of 3). As a result, 2 is ineffective.
When you use 3 O2, you have 6 oxygen atoms on the reactant side and 6 oxygen atoms on the product side. This is effective! The balanced chemical equation is as follows:
4 Fe + 3 O2 → 2 Fe2O3
Chemists, on the other hand, always write the simplest equation, so double-check your work to ensure you can’t lower your coefficients.
This is how a simple chemical equation for mass is balanced. You may also need to balance mass and charge equations. You may also need to specify the state of matter (solid, liquid, aqueous, or gas) of the reactants and products.
Examples of Representation of Chemical Reactions
- Solid calcium carbonate is heated and decomposes to solid calcium oxide and carbon dioxide gas.
CaCO3 → CaO + CO2
- Water vapour reacts with sodium metal to produce solid sodium hydroxide and hydrogen gas.
2H2O + 2Na → 2NaOH + H2
- Aqueous solutions of magnesium chloride and sodium hydroxide react to produce solid magnesium hydroxide and aqueous sodium chloride.
MgCl2 + 2NaOH → 2NaCl + Mg(OH)2
- The reaction of molecular nitrogen (N2) and oxygen (O2) to form dinitrogen pentoxide.
2N2 + 5O2 → 2N2O5
- Phosphorus burns in presence of chlorine to form phosphorus pentachloride.
P4 + 10Cl2 → 4PCl5
- The process of respiration.
C6H12O6 + 6O2 + 6H2O → 6CO2 + 12H2O + energy
Examples of Balanced Equations
Balance the following equation:
- HNO3 +P2O5 → N2O5 + HPO3
Balanced Equation: 4HNO3 +P2O5 → 2N2O5 + HPO3
- HNO +Ca(OH)2 → Ca(NO3)2 + H2O
Balanced Equation: 2HNO3 + Ca(OH)2 → Ca(NO3)2 + 2H2O
- BaCl2 +H2SO4 → BaSO4 +HCl
Balanced Equation: BaCl2 +H2SO4 → BaSO4 + 2HCl
- NaCl + AgNO3 → AgCl + NaNO3
Balanced Equation: NaCl + AgNO3 → AgCl + NaNO3
References
- https://www.inspiritvr.com/general-chemistry/chemical-reactions/what-does-an-arrow-mean-in-a-chemical-equation-study-guide
- James E. Brady; Frederick Senese; Neil D. Jespersen (2007). Chemistry: Matter and Its Changes. John Wiley & Sons. ISBN 9780470120941.
- Thorne, Lawrence R. (2010). “An Innovative Approach to Balancing Chemical-Reaction Equations: A Simplified Matrix-Inversion Technique for Determining the Matrix Null Space”. Chem. Educator. 15: 304–308.
- https://www.ck12.org/c/chemistry/reactants-and-products/lesson/Reactants-and-Products-CHEM/
- Experimental Data and Empirical Formulas. https://courses.lumenlearning.com/boundless-chemistry/chapter/experimental-data-and-empirical-formulas/
- https://dept.harpercollege.edu/chemistry/chm/100/dgodambe/thedisk/chemrxn/rxnintr2.htm
- Nivaldo Tro, Travis Fridgen, Lawton Shaw. Chemistry a Molecular Approach. 3rd ed. 2017.Final Representing
- https://water.mecc.edu/courses/Env211/lesson6_2b.htm
- https://unacademy.com/content/nda/study-material/chemistry/symbolic-representation-of-a-chemical-equation/
- Helmenstine, Anne Marie, Ph.D. “How to Balance Chemical Equations.” ThoughtCo, Apr. 5, 2023, thoughtco.com/how-to-balance-chemical-equations-603860.
