Roentgenium is a synthetic transition metal with an atomic number of 109 and is represented by the symbol ‘Rg’ in the periodic table. It is silvery in appearance and belongs to the d-block of period 7 of the periodic table. Roentgenium was the fifth transactinide (super-heavy) element identified. Only tiny quantities of Roentgenium have been successfully synthesized, hence there isn’t much known about it based on experimental data, but some qualities can be predicted using periodic table trends.
Roentgenium, a super-heavy radioactive element that does not occur naturally, is produced inside a laboratory setting and decays within milliseconds after being synthesized. In December of 1994, a group of researchers supervised by Sigurd Hofmann at the GSI Helmholtz Centre for Heavy Ion Research in Germany successfully synthesized roentgenium. The name roentgenium was officially assigned in honor of the achievements of physicist Wilhelm Rontgen who identified the X-rays.
Discovery and History of Roentgenium
- The Joint Institute for Nuclear Research (JINR) tried blasting bismuth with nickel in an effort to synthesize element-111, however they were unable to find any element-111 atoms.
- In December of 1994, a group of researchers supervised by Sigurd Hofmann at the GSI Helmholtz Centre for Heavy Ion Research in Germany successfully synthesized roentgenium.
- Following a nickel bombardment of bismuth, they were able to produce a small number of atoms of isotope-272 of element 111, which had a half-life of 1.5 milliseconds.
- Three more atoms of roentgenium-272 were created in 2002 using the same methodology.
- In 2004, element-111 was officially renamed to “Roentgenium,” following GSI’s suggestion.
- In recognition of the work done by physicist Wilhelm Rontgen, who discovered X-rays, the element known as roentgenium was named after him.
Occurrence of Roentgenium
- Roentgenium can be synthesized artificially. It’s a synthetic element that is extremely unstable. Its half-life is only a few seconds.
- Roentgenium is a synthetic radioactive metal formed by nuclear bombardment and has only been manufactured in trace amounts.
- Roentgenium is created by blasting 209Bi with 64Ni in a heavy ion accelerator.
- There are a total of seven isotopes of Roentgenium with mass numbers ranging from 272 to 282, whose half-lives are known.
Elemental Properties of Roentgenium
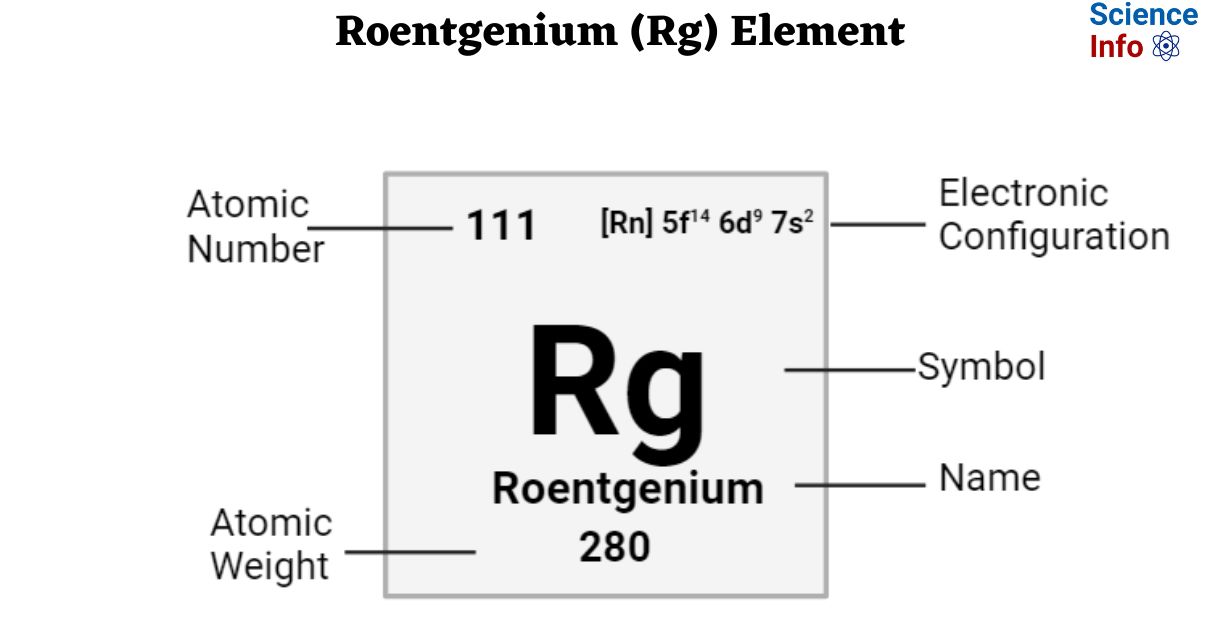
| Electronic Configuration | [Rn] 5f14 6d9 7s2 |
| Atomic Number | 111 |
| Atomic Weight | 280 g.mol -1 |
| State at 20°C | Solid |
| Group, Period, and Block | Trans-actinides, 7, d-block |
| Density | 28.80 g/cm3 (estimated) |
| Ionic radius | – |
| Van der Waals radius | – |
| Electron shells | 2, 8, 18, 32, 32, 17, 2 (estimated) |
| Electrons | 111 |
| Protons | 111 |
| Neutrons | 170 |
Isotopic Information of Roentgenium
- Roentgenium has no stable isotopes naturally, but they can be created in a laboratory setting.
- All of the Roentgenium isotope are unstable and radioactive.
- All isotopes of Roentgenium decay via alpha decay or spontaneous fission, but none of them undergo beta decay.
- It contains nine isotopes with known half-lives: 272Rg, 274Rg, 278Rg, 279Rg, 280Rg, 281Rg, 282Rg, 283Rg, and 286Rg.
- Isotopes are created when a heavier element decays or when two light nuclei fuse together.
- The heavier isotopes are more stable than the lighter ones. The most stable and widely recognized roentgenium isotope is 282Rg, and is also the most heavy documented isotope, with a half-life of 2 minutes and 10 seconds.
- The unverified 286Rg is predicted to be heavier and have a longer half-life of around 10.7 minutes, which makes it the longest-lived super-heavy known nuclide.
- Similarly, the unverified 283Rg expected to have a long half-life of 5.1 minutes.
- The isotopes 281Rg and 280Rg have also been reported to have half-lives greater than one second. Furthermore, the remaining isotopes have half-lives of a millisecond
Physical Properties of Roentgenium
- Roentgenium’s volatility makes it difficult to perform a statistically significant study of its physical properties.
- Due to its rapid disintegration, only few properties of Roentgenium have been investigated until now.
- Roentgenium is a synthetic, super-heavy transactinide element. It is expected to be a solid under normal conditions.
- It is projected that all elements from 104 to 111 will display fourth transition metal characteristics. This series also includes Roentgenium, which is a platinum group metal.
- It is found in the 7th period, the 11th Group, and the d-block of the periodic table.
- It is silvery in appearance. It is projected to be silver-colored based on the difference between the ground state and the first excited state of the outer d-electrons.
- If enough element 111 is ever synthesized, it will most likely be softer than gold. Rg+ is expected to be the softest of all metal ions.
- The melting point and the boiling point of the element 111 is yet to be known.
- The atomic mass of Roentgenium is 282. The atomic mass of man-made trans-uranium elements is calculated using the periodic table’s longest-lived isotope. These atomic weights should be considered tentative because a new isotope with a longer half-life may be created in the future.
- Roentgenium is expected to be a solid under normal conditions. It is projected to have an high density of around 28.80 g/cm3.
- In contrast to lighter elements, which have face-centered cubic crystal structures, Rg is expected to form body-centered cubic crystals. This is due to the differential in electron charge density of roentgenium.
- Apart from nuclear properties, no physical features of Roentgenium have been researched so far due to the short half-life of all of its isotope and the high cost of production.
- The most stable roentgenium isotope is 282Rg, with a half-life of 2 minutes and 10 seconds.
Chemical Properties of Roentgenium
- Roentgenium is an extremely radioactive element. Roentgenium’s chemical characteristics have yet to be thoroughly researched. Isotopes have short half-lives, and the molecules they contain are extremely volatile, making statistically meaningful chemical analysis difficult.
- There have been no experimental measurements of Roentgenium compounds, and all known predictions are theoretical.
- Roentgenium is projected to be a noble metal. (The chemical elements that exist in solid metal form, are exceptionally resistant to oxidation and high temperatures, have anti-corrosive properties, and do not react strongly with acids.)
- Based on the oxidation states of other member of its group (Group 11), the common oxidation states of roentgenium is predicted to be +5 and +3.
- Roentgenium compounds are expected to have a stable state of +3.
- It is anticipated to create more diverse and steady compounds, but its reactivity is comparable to that of gold.
- It has also been anticipated that Roentgenium will combine with phosphine, ammonia, as well as hydrogen to produce compounds.
Synthesis of Roentgenium
- All elements with atomic numbers more than 100 can only be created through reactions in a particle accelerator, such as a cyclotron; they do not develop in a nuclear reactor.
- When nickel-64 bombards bismuth-209, roentgenium-272 is produced.
Uses of Roentgenium
- Given barely any atoms of this metal have been created to date, there are currently no specific or exclusive uses of Roentgenium outside of scientific research.
- Furthermore, because it is unavailable in nature, Roentgenium is only employed by scientific researchers, with no recognized negative effects or uses for the metal among individuals and organizations
- A consistent scientific experiment intended to deliver an evident outcome requires a large number of atoms of the same element. However, only few atoms of Roentgenium have been synthesized so far.
Health Effects of Roentgenium
- Roentgenium is a very unstable chemical; when created, it swiftly decomposes into other elements, therefore it has no impact on human health.
Environmental Effects of Roentgenium
- Roentgenium’s environmental effects are negligible due to its short half-life (just a few seconds).
Video Reference
References
- https://periodic-table.com/roentgenium/
- https://www.chemicool.com/elements/roentgenium.html
- https://www.thoughtco.com/roentgenium-facts-rg-or-element-111-3876744
- https://chemicalengineeringworld.com/roentgenium-element-properties-and-information/
- https://www.azom.com/article.aspx?ArticleID=7951https://www.livescience.com/41347-facts-about-roentgenium.html
- https://www.vedantu.com/chemistry/roentgenium
