Europium is a chemical element with an atomic number of 63 and is represented by the symbol ‘Eu’ in the periodic table. It is hard and silvery in appearance classified as a rare earth metal and belongs to the f-block of the lanthanide group of the periodic table. Europium commonly manifests an oxidation state of +3, similar to other elements within the lanthanide series. Nevertheless, it is important to acknowledge that compounds exhibiting an oxidation state of +2 are also commonly observed. Europium compounds with an oxidation state of +2 demonstrate a moderately reducing character.
Europium demonstrates the highest chemical reactivity, lowest density, and greatest softness among the elements in the lanthanide series. Europium is categorized as one of the scarcest rare-earth elements present on Earth. Europium does not exist in its elemental form in nature.
History of Europium
- The element europium was first discovered in 1896 by Eugène-Antole Demarçay, a renowned chemist from France.
- Demarçay had suspicions that the samples of samarium, a newly discovered element, might have been tainted with an unidentified element.
- In 1901, he achieved the remarkable feat of isolating europium by employing a series of crystallization techniques on samarium magnesium nitrate.
- The process of isolating europium from impure gadolinium was conducted in 1904 by French chemist Georges Urbain, who employed bismuth magnesium nitrate for this purpose.
- Europium gets its name from the continent of Europe.
Occurrence of Europium
- Europium is classified as one of the most limited rare-earth elements found on the planet. Europium is not naturally present in its elemental state within the environment.
- Europium is not naturally occurring in its free form but instead is commonly found within various minerals, predominantly monazite, bastnaesite, and xenotime.
- In commercial practice, the isolation of europium is achieved through the utilization of ion exchange and solvent extraction techniques. The production of pure metal can be achieved through the process of electrolysis, wherein the molten chloride is subjected to sodium chloride.
- Europium exhibits a total of 30 isotopes, each possessing a distinct half-life, ranging from mass numbers 131Eu to 162Eu. Europium found in nature is a composite of its two stable isotopes.
Isotopes of Europium
Europium has two naturally occurring isotopes: 151 Eu and 153 Eu.
Naturally Occurring Isotopes of Europium
| Isotopes | Natural Abundance (atom %) |
|---|---|
| 151 Eu | 47.8 |
| 153 Eu | 52.2 |
Elemental Properties of Europium
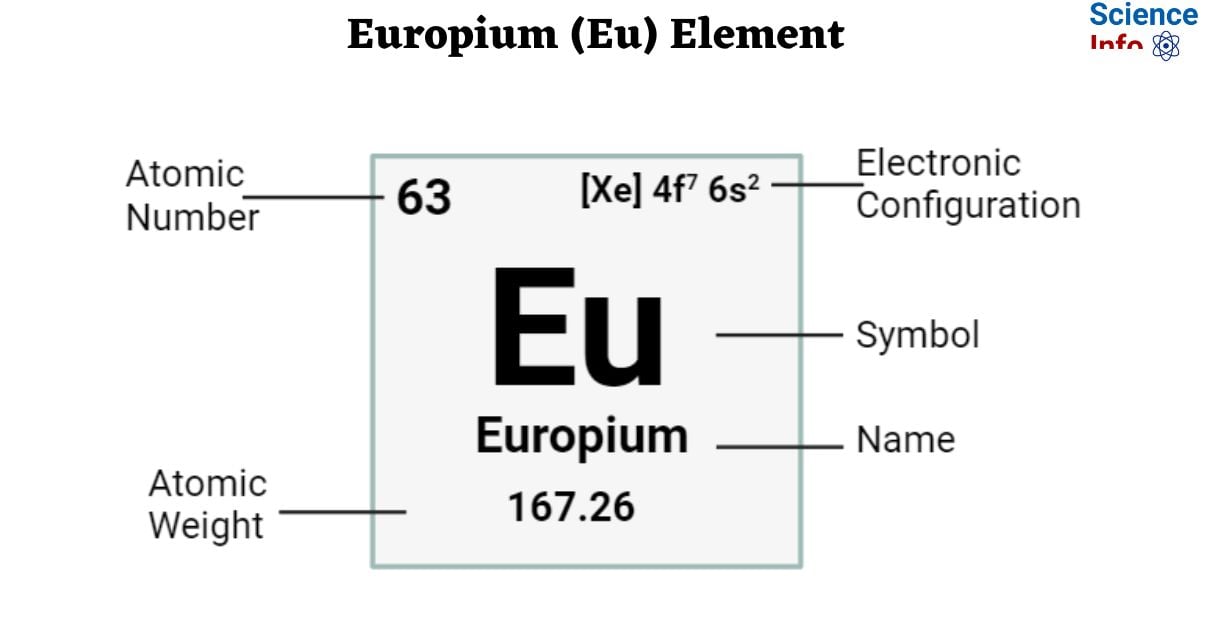
| Electronic Configuration | [Xe] 4f7 6s2 |
| Atomic Number | 63 |
| Atomic Weight | 167.26 g.mol -1 |
| State at 20°C | Solid |
| Group, Period, and Block | Lanthanides, 6, f-block |
| Density | 5.248 g/cm3 at 20 °C |
| Appearance | silvery-white |
| Van der Waals radius | unknown |
| Electron shells | 2, 8, 18, 25, 8, 2 |
| Electrons | 63 |
| Protons | 63 |
| Neutrons in the most abundant isotope | 90 |
Physical Properties of Europium

- Europium has an atomic number of 63 and is a silvery-white rare earth metal. It has a melting point of 826°C (1520°F) and a boiling point of 1489°C (2712°F).
- Eu has a solid phase density of 5.244 g/cm3 and a liquid or molten phase density of 5.13 g/cm3.
- It is malleable which means it can be easily beaten into thin sheets without any cleavage.
- It is ductile which means it is possible to draw thin wires from it without breaking.
- The body-centered-cubic structure can be seen in europium crystals.
- Europium typically exists in a divalent state under normal conditions. The application of pressure results in the conversion of the substance into a trivalent state. As a result of this alteration, europium exhibits the characteristics of a superconductor.
| Color/physical appearance | metallic, silvery-white |
| Melting point/freezing point | 1099 K (826 °C, 1519 °F) |
| Boiling point | 1802 K (1529 °C, 2784 °F) |
| Density | 5.24 g cm-3 at 20° |
| Malleability | Yes |
| Ductility | Yes |
| Electronegativity | 1.2 (Pauling Scale) |
Chemical Properties of Europium
- Europium exhibits the highest reactivity compared to other elements in the lanthanide series. When exposed to water, it undergoes a rapid reaction that results in the release of hydrogen gas. Additionally, it exhibits a highly reactive nature when exposed to atmospheric oxygen, resulting in spontaneous combustion.
- Europium exhibits a pronounced reactivity with atmospheric oxygen, leading to spontaneous combustion. Consequently, it necessitates storage within an inert atmosphere to impede oxidation.
- Europium demonstrates a relatively low reactivity when exposed to cold water, whereas its reactivity is enhanced when exposed to hot water, leading to the formation of a hydroxide compound.
- Europium compounds have been shown to exhibit a +3 oxidation state, similar to other lanthanides. However, it is noteworthy that compounds with a +2 oxidation state are as well commonly observed.
- Europium exhibits reactivity with halogens, resulting in the formation of trihalides.
- Europium exhibits the ability to form chalcogenides and pnictides that possess notable stability.
Chemical Reaction of Europium
- The Reaction of Europium With Air
Europium exhibits a gradual process of oxidation when exposed to atmospheric conditions, resulting in the formation of europium (III) oxide, denoted as Eu2O3, which readily combusts.
4 Eu (s) + 3 O2 (g) → 2 Eu2O3 (s)
- The Reaction of Europium With Water
Europium exhibits a sluggish reaction rate when exposed to cold water, whereas it undergoes a rapid reaction when in contact with hot water. This reaction results in the formation of europium hydroxide, denoted as Eu(OH)3, along with the liberation of hydrogen gas (H2).
2 Eu (s) + 6 H2O (g) → 2 Eu(OH)3 (aq) + 3 H2 (g)
- The Reaction of Europium With Halogens
The element europium exhibits a propensity to engage in chemical reactions with various halogens, resulting in the formation of europium (III) halides.
The chemical reaction between europium metal and fluorine gas (F2) results in the formation of europium (III) fluoride, denoted as EuF3.
2 Eu (s) + 3 F2 (g) → 2 EuF3 (s) [white]
The chemical reaction between europium metal and chlorine gas (Cl2) results in the formation of europium (III) chloride, denoted as EuCl3.
2 Eu (s) + 3 Cl2 (g) → 2 EuCl3 (s) [yellow]
The chemical reaction between europium metal and bromine (Br2) results in the formation of europium (III) bromide, denoted as EuBr3.
2 Eu (s) + 3 Br2 (g) → 2 EuBr3 (s) [grey]
The chemical reaction between europium metal and iodine represented as I2, results in the formation of europium (III) iodide, denoted as EuI3.
2 Eu (s) + 3 I2 (g) → 2 EuI3 (s)
- The Reaction of Europium With Acid
The dissolution of europium in dilute sulfuric acid is characterized by its high reactivity, resulting in the formation of Eu (III) ions and the liberation of hydrogen gas, denoted as H2. The solution exhibits a pale pink hue, indicating a high probability of being attributed to the complex [Eu(H2O)9]3+.
2 Eu (s) + 3 H2SO4 (aq) → 2 Eu3+ (aq) + 3 SO42− (aq) + 3 H2 (g)
Uses of Europium
There are just a few applications for Europium in various sectors. Some of the important applications of europium are discussed here:
- Europium serves as a dopant material in certain glass compositions, particularly in lasers and various optoelectronic applications.
- Europium oxide is commonly employed as a red phosphor in television tubes due to its ability to generate a vibrant red color. Yttrium phosphors can be activated using it as well.
- Europium is employed in quantum memory chips, which serve the purpose of securely retaining quantum data for a limited duration within a portable, disk-shaped apparatus, facilitating its transportability.
- The Europium isotope’s mass number facilitates its efficient absorption of neutrons without inducing any radioactive reactions. This is the rationale behind its utilization as a control rod within a nuclear reactor.
- Europium is present in a minor quantity within the luminous fixtures employed for street lighting. The aforementioned element is also found in mercury vapor lamps, which are utilized for the emission of natural light.
- Europium exhibits a notable neutron capture cross-section and possesses properties that render it potentially valuable as a neutron poison in the event of a thermo-nuclear hazard.
- Europium-doped phosphors serve as fluorescent security markers on Euro banknotes, exhibiting a red luminescence when exposed to ultraviolet (UV) light. Consequently, these markers aid in the identification and prevention of counterfeit currency.
- Plastics doped with europium serve as laser materials and are employed in the production of thin superconducting alloys.
Health Effects of Europium
The biological function of europium is unknown. Although its toxicity has not been thoroughly studied, ingesting europium salts may be somewhat hazardous.
Environmental Effects of Europium
No plants, animals, or environmental factors are endangered by europium. Explosive or fire risks are created by the metal particles.
References
- https://www.britannica.com/science/europium
- https://www.rsc.org/periodic-table/element/63/europium
- https://pubchem.ncbi.nlm.nih.gov/element/Europium
- Ferenc Szabadváry, Handbook of the Chemistry and Physics of the Rare Earths Vol. 11., Elsevier Science Publishers., 1998, p65.
- Per Enghag, Encyclopedia of the elements: technical data, history, processing, applications., John Wiley and Sons, 2004, page 450.
- https://www.lenntech.com/periodic/elements/eu.htm
- John Emsley, Nature’s building blocks: an A-Z guide to the elements., Oxford University Press, 2003, p140.
- https://www.chemicool.com/elements/europium.html
- https://byjus.com/chemistry/europium/
- https://education.jlab.org/itselemental/ele063.html
