Glycolysis is an oxidative process that converts one mole of glucose into two moles of pyruvate through a series of enzymatic reactions. It is also known as the Embden-Meyerhof pathway.
The pyruvate obtained from the glycolysis process further enters into the citric acid cycle or Kerb’s cycle. It is a major pathway for glucose metabolism and occurs in the cytosol of cells. It is a distinct pathway that occurs both aerobically and anaerobically and does not require molecular oxygen. Glycolysis is the fundamental process that underpins both aerobic and anaerobic cellular respiration.
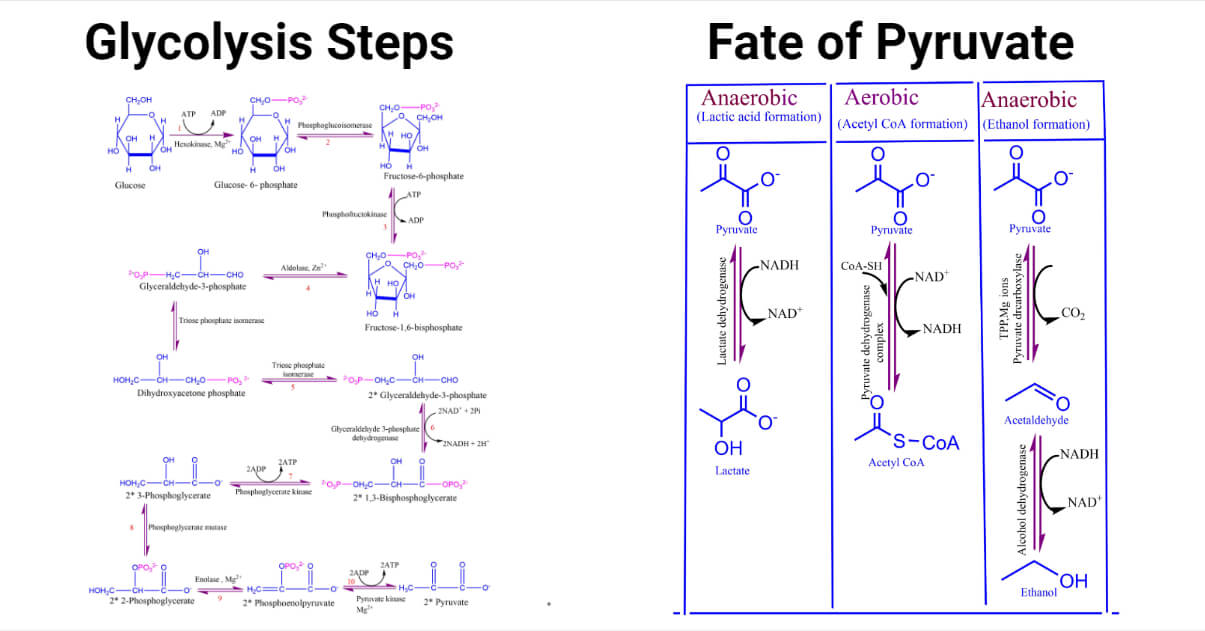
Step of Glycolysis
1. Phosphorylation (1st step)
Glucose is phosphorylated by ATP to form a phosphate sugar, glucose 6-phosphate. The negative charge of phosphate prevents sugar-phosphate from passing through the plasma and traps glucose within the cell. It is an irreversible reaction catalyzed by the enzyme hexokinase.
2. Isomerization (2nd step)
The isomerization of glucose-6-phosphate to fructose-6-phosphate converts aldose to ketose. In this step, the reversible arrangement of the carbonyl group from carbon 1 to carbon 2 takes place by forming ketose from aldose sugar. THs step is catalyzed by the enzyme phosphoglucoisomerase.
3. Phosphorylation (3rd step)
The fructose-6-phosphate is phosphorylated by ATP to form fructose 1,3-bisphosphate. Bisphosphate indicates that two monophosphate groups are present separately. The enzyme phosphofructokinase catalyzes this reaction step.
4. Cleavage (4t step)
The six-carbon sugar cleaved to form three carbon molecules glyceraldehyde-3-phosphate and dihydroxyacetone phosphate. Aldolase catalyzes this reaction.
5. Isomerization (5th step)
Dihydroxyacetone phosphate is isomerized to form glyceraldehyde-3- phosphate. This reaction is catalyzed by the enzyme triose phosphate isomerase.
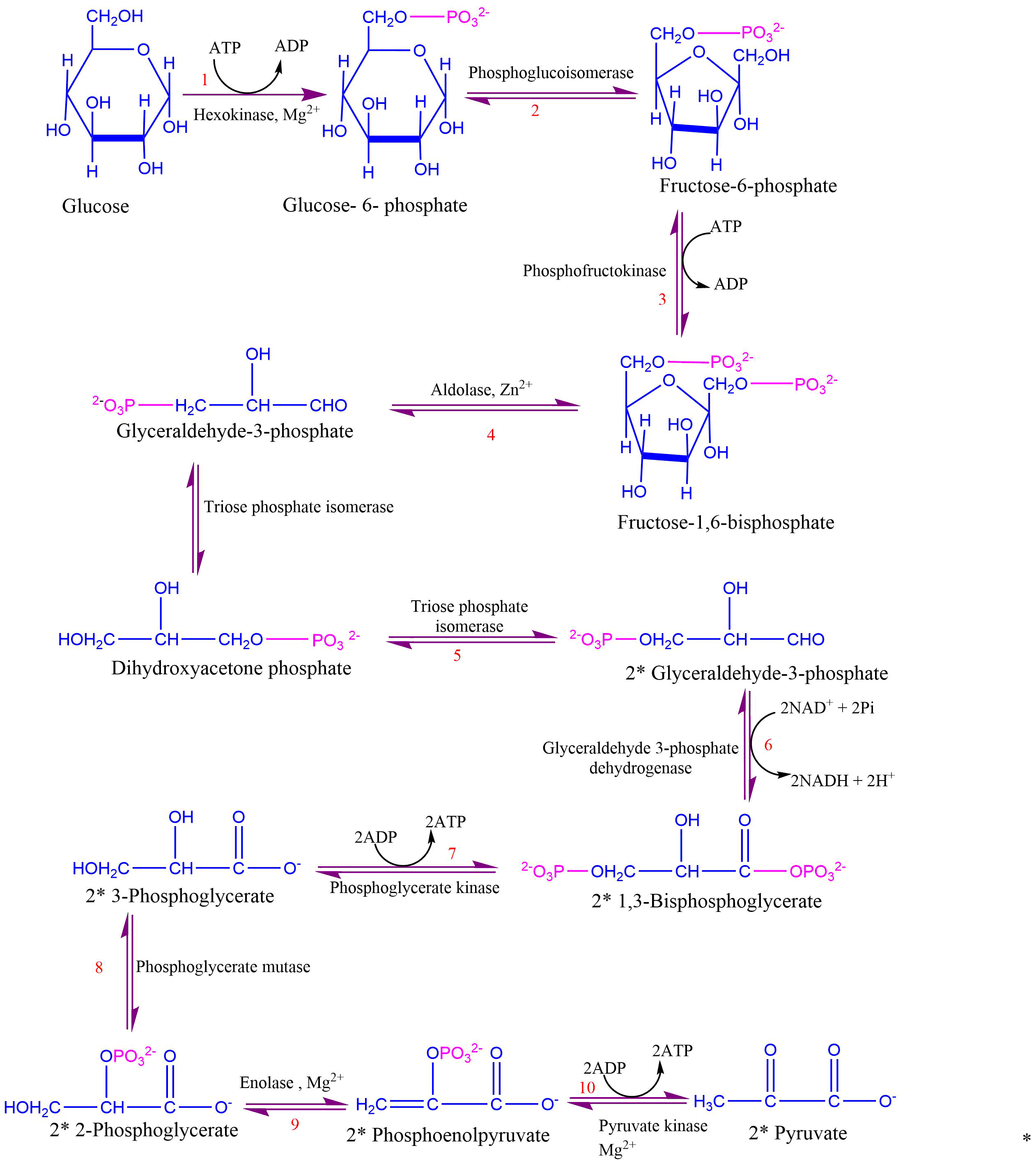
6. Oxidation (6th step)
Two molecules of glyceraldehyde -3-phosphate is oxidized and converted into 1,3-bisphosphoglycerate. Enzyme glyceraldehyde 3-phosphate dehydrogenase catalyzes this reaction.
7. Phosphorylation (7th step)
In this step, the high-energy phosphate group from step 6 is transferred from ADP to form ATP. As the phosphate donor, 1,3 bisphosphoglycerate, is the substrate with high phosphoryl transfer potential, this ATP formation is known as substrate level phosphorylation.
8. Rearrangement (8th step)
The phosphate ester linkage in 3-phosphoglycerate is moved from carbon 3 to carbon 2 to form 2-phosphoglycerate.
9. Dehydration (9th step)
Removal of water from 2-phosphoglycerate results in the formation of high-energy enol phosphate linkage. Enolase catalyzes this reaction.
10. Transfer of phosphoryl group (10th step)
It is the last step of glycolysis that involves the irreversible transfer of phosphoryl group from phosphoenolpyruvate to ADP. Enzyme pyruvate kinase catalyzes this reaction.
Energy in Glycolysis
The energy yield from the glycolysis is 2ATP and 2NADH. In the presence of oxygen, one molecule of NADH oxidized to yield 2.5 ATP. So, two molecules of NADH produce 2*2.5 = 5 ATP. The aerobic glycolysis produces 7 ATP (5 ATP from oxidation of NADH, 2 ATP from substrate level phosphorylation). Glycolysis produces 2 ATP as a result of substrate-level phosphorylation in absence of oxygen (anaerobic glycolysis).
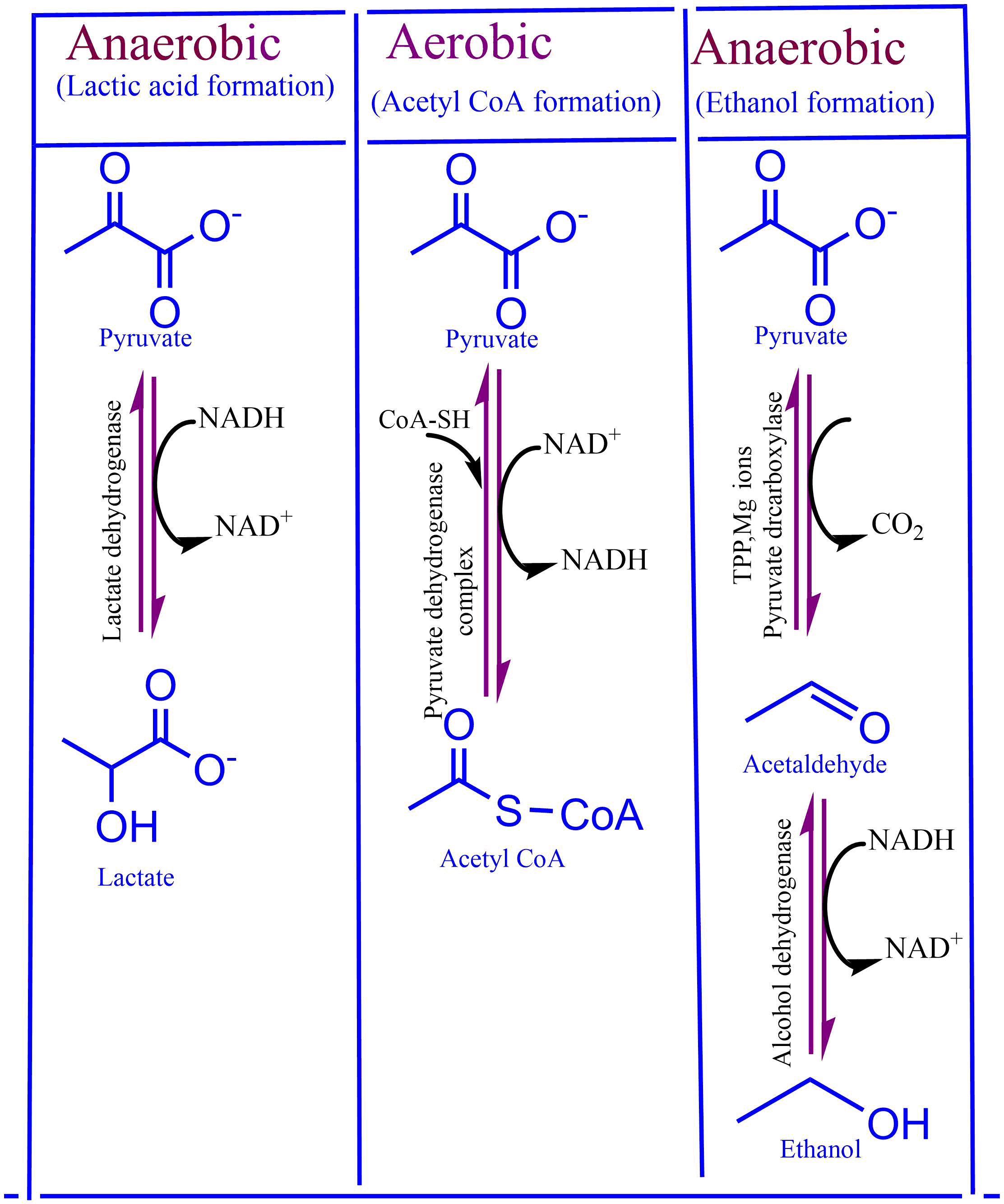
Fate of Pyruvate
Depending on the type of cell that produces it, pyruvate has a catabolic fate. If the cells that produce pyruvate are aerobic, then pyruvate further will convert to acetyl-CoA following the aerobic pathway. Similarly, in the presence of hypoxia in cells (lactic acid bacteria, skeletal muscles, plants, etc.), pyruvate must undergo anaerobic metabolism, resulting in the formation of lactate or ethanol.
References
- https://www.britannica.com/science/glycolysis
- https://byjus.com/biology/glycolysis/#:~:text=Glycolysis%20is%20the%20process%20in,both%20aerobic%20and%20anaerobic%20organisms.
- https://www.khanacademy.org/science/biology/cellular-respiration-and-fermentation/glycolysis/a/glycolysis
- https://microbiologyinfo.com/glycolysis-10-steps-explained-steps-by-steps-with-diagram
- https://www.vedantu.com/biology/glycolysis
- https://thebiologynotes.com/fate-of-pyruvate/
- https://microbeonline.com/fate-of-pyruvate/
