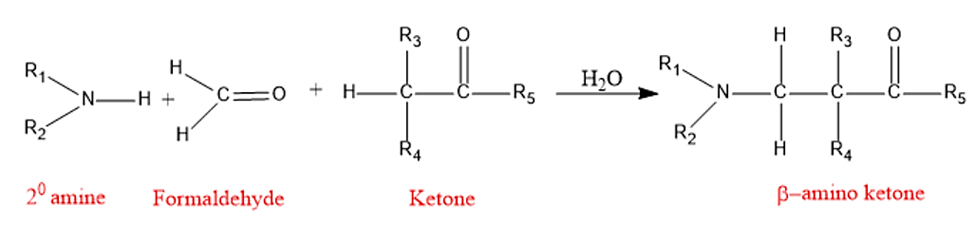
The Mannich reaction is a fundamental and significant C-C bond formation process in organic synthesis. The Mannich reaction is an organic reaction that is used to create a β-amino carbonyl molecule from a primary or secondary amine and two carbonyl compounds (one non-enolizable and one enolizable).
Mannich reaction was named after Carl Ulrich Franz Mannich, a German chemist who discovered it in 1912.
What is Mannich Reaction?
The Mannich reaction is an amino alkylation reaction that involves the condensation of an enolizable carbonyl compound (α-CH acidic compound) with a nonenolizable aldehyde (like formaldehyde) and ammonia; or a primary or secondary amine to yield a β-aminocarbonyl compound, also known as Mannich base.
The Mannich reaction is an organic reaction that consists of a formaldehyde and ammonia or any primary or secondary amine amino alkylation of an acidic proton next to a carbonyl functional group to form β-amino-carbonyl compound. Aldimines form between amines and aldehydes, therefore reactions between them and α-methylene carbonyls are also regarded as Mannich reactions.
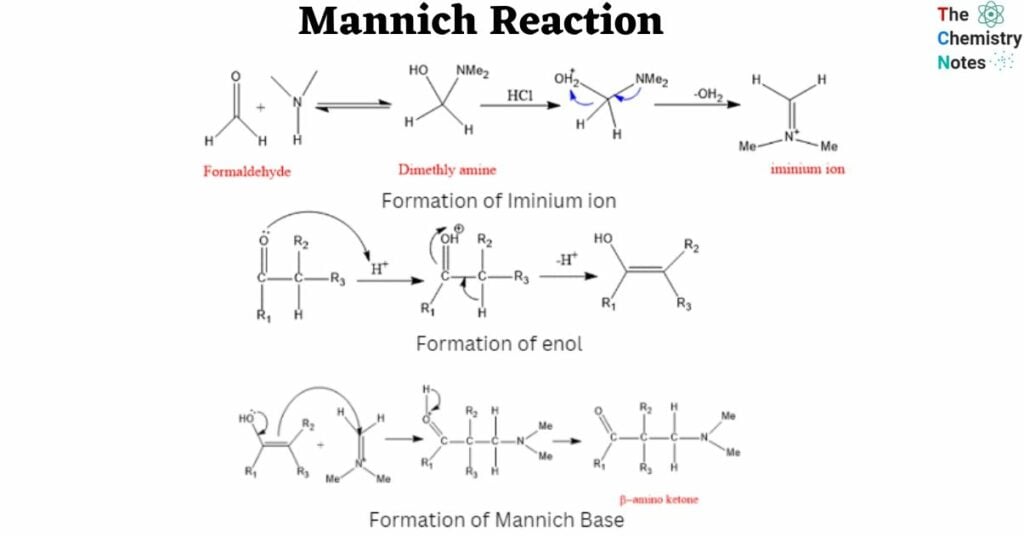
Mechanism of Mannich Reaction
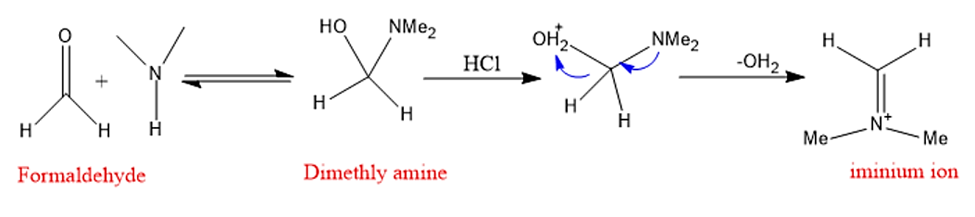
Step 1: In the first stage, formaldehyde combines with amine, which is then deprotonated and dehydrated to create iminium ion.
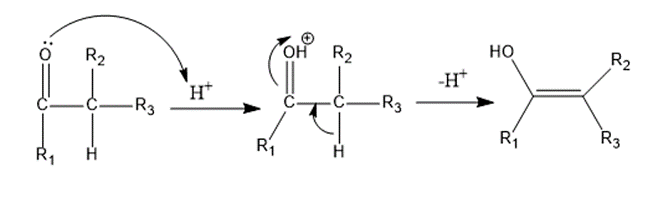
Step 2: In the second step, the carbonyl molecule (ketone in this case) with an α-hydrogen is protonated and subsequently deprotonated to undergo tautomerization and generate an enol form.
Step 3: The required product, β-amino carbonyl compound, is synthesized after deprotonation of the compound produced by this enol, the nucleophile, attacking the iminium ion.
![Formation of Mannich Base [Mannich reaction]](https://scienceinfo.com/wp-content/uploads/2023/08/image-157.png)
Applications of Mannich Reaction
- This reaction is used for the preparation of alkyl amines which are used for making agrochemicals such as pesticides.
- It is used for the synthesis of organic compounds such as nucleotides and peptides.
- Mannich bases are widely used in the pharma industry for the synthesis of numerous pharmaceutical drugs such as fluoxetine, which is a strong antidepressant.
- Catalysts and polymers are made by the Mannich reaction.
- A lot of antibiotics are Mannich bases. Tetracycline is an antibiotic whose Mannich base is rolitetracycline.
- Using Mannich Reaction non-polar hydrocarbons can be converted into soaps and detergents.
- Cleaning solutions, vehicle fuel treatment, and epoxy coatings are just a few of the applications. Substituted long-chain alkyl ethers are converted to polyether amines by a variety of techniques.
- Tropinone is synthesized by the Robinson-Schopf process, which includes the Mannich reaction. Tropinone is produced by treating succindialdehyde (butanedial) with methylamine and 3-oxoglutaric acid (3-oxopentanedioic acid).
Video on Mannich Reaction
References
- Morrison R. T. & Boyd R. N. (1983). Organic chemistry (4th ed.). Allyn and Bacon.
- Smith M. & March J. (2001). March’s Advanced Organic Chemistry: Reactions Mechanisms and Structure (5th ed.). Wiley.
- Ghosh, S.K., Advanced General Organic Chemistry, Second Edition, New Central Book Agency Pvt. Ltd., Kolkatta, 2007.
- https://www.organic-chemistry.org/namedreactions/mannich-reaction.shtm
- https://byjus.com/chemistry/mannich-reaction-mechanism/
