The Nuclear Overhauser Effect (NOE) is described as the change in intensity of one NMR resonance caused by the saturation of another. It is caused by dipole-dipole cross-relaxation between nuclei, and its utility derives from the fact that the strength of a given NOE enhancement is roughly associated with internuclear separation.
The NOE in tiny compounds in solution is positive (it causes impacted resonances to intensify) and is often studied using one-dimensional experiments. The NOE is negative in larger molecules (affected resonances lose intensity) and is often assessed using the two-dimensional NOESY experiment or one of its multidimensional versions.
What is the Nuclear Overhauser Effect?
Overhauser first described the Nuclear Overhauser Effect as a strategy for greatly increasing the intensity of NMR signals by irradiating some of the nearby nuclei within the molecule.
The Nuclear Overhauser effect (NOE) is a “through space” effect that can be seen by irradiating a peak in the sample that corresponds to a specific proton with lower power radiation than is utilized for decoupling.
The Nuclear Overhauser effect (NOE effect) is the phenomenon that occurs when a proton peak is irradiated at its resonance frequency, causing an increase in the peak intensity of a proton that is close in space to that irradiated proton.
Explanation for Nuclear Overhauser Effect?
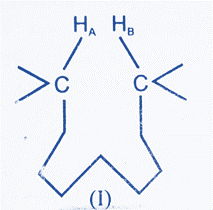
Consider two distinct protons, HA and HB, in the hypothetical molecule (I), which are too close for any through-space contact of their fluctuating magnetic vectors; one can contribute to the spin-lattice relaxation process of the other. There are too many intervening links between HA and HB for any through-bond interaction to occur.
The compound’s spectra are run and integrated. It is then reintegrated with a rather low-power decoupler irradiating HB. If HA and HB are reasonably close in space (i.e., less than 4.5Å apart), the intensity of HA will be slightly increased in the second spectrum. No such increase will be detected if HA and HB are separated by more than 4.5 Å.
The nuclear overhauser effect is very useful in studying the molecular geometry of compounds because it tells whether the two protons are in close proximity to the molecules or not. An important consequence of this effect is that the line intensities observed in the normal spectrum may not be the same as those observed in the decoupled spectrum.
Significance and Applications of Nuclear Overhauser Effect
- Organic chemists may want to know whether the substituents in numerous bicyclic compounds are in the exo or endo position. Many of these difficulties cannot be handled using chemical shift analysis or the spin-spin coupling phenomenon. NOE difference spectroscopy is quite effective in resolving these issues.
- A typical 1H-NMR peak is removed from a specific proton irradiation spectrum in the NOE difference experiment. As a result, we receive a spectrum that contains only the peaks that have been amplified by the NOE effect.
- The NOE experiment is extremely useful for distinguishing protons based on their spatial locations. In alkenes, for example, it is possible to detect whether the groups are cis or trans.
Applications of the Nuclear Overhauser Effect
- In NMR spectroscopy, the NOE is useful for determining which protons are close together in space. NOE can be used to indicate that two protons or groups of protons exist in close proximity in space within a molecule by properly measuring it. As a result, it has useful applications in structural elucidation, molecular geometry determination, and stereochemistry.
- Its applications have grown significantly, including conformational analysis, intermolecular interaction measurement, and ligand binding study.
- It is especially effective in small molecules for determining specific stereochemical interactions, such as substitution or ring fusion patterns in mostly rigid systems.
- It can establish precise three-dimensional solution structures for macromolecular systems such as proteins and their complexes with DNA, RNA, or other proteins.
References
- https://chem.libretexts.org/Ancillary_Materials/Worksheets/Spectroscopy_(Worksheets)/22%3A_Nuclear_Overhauser_Effect_(NOE)
- https://link.springer.com/chapter/10.1007/1-4020-3910-7_50
- https://www.sciencedirect.com/topics/medicine-and-dentistry/nuclear-overhauser-effect.
- https://www.slideshare.net/grakbph040/nuclear-overhauser-effect
