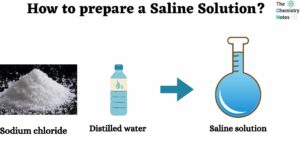
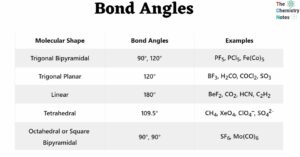
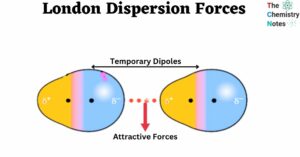
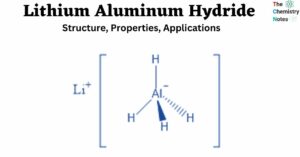
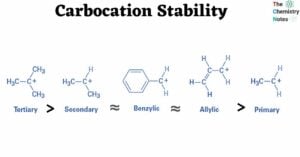
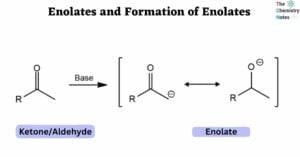
Solubility Rules
When a material is combined with a solvent, various outcomes are possible. The substance’s solubility, which is defined as the solute’s highest achievable concentration, is what determines the outcome. The solubility rules aid in determining which compounds are soluble and … Read more