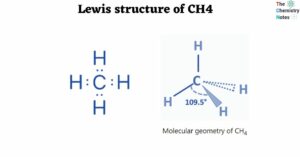
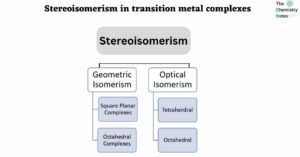
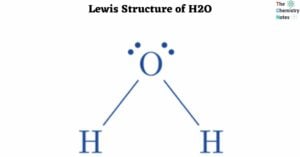
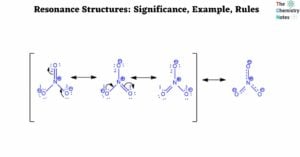
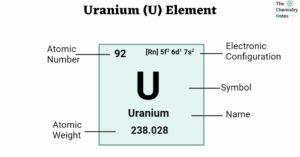
Lewis Structure of CH4
The Lewis structure of a molecule depicts the arrangement of atoms and valence electrons. The Lewis structure of CH4 is a basic but important as it helps us understand the form and chemical activity of the molecule by highlighting the electron … Read more