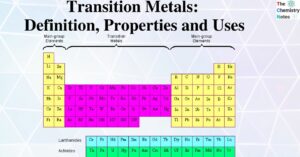
Qualitative Salt Analysis: Common Ion Effect
Qualitative salt analysis is the process of determining the number of constituents present in an unknown salt sample. The common ion effect is most commonly used in the salt analysis, i.e. qualitative analysis of basic radicals (in group separation). In qualitative … Read more