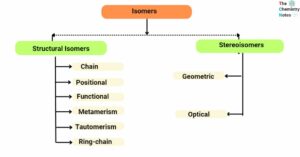
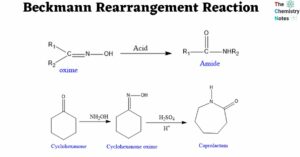
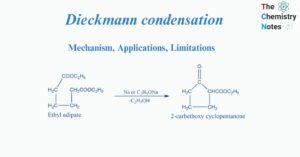
Zaitsev’s rule: Definition, Applications, Limitations
Zaitsev’s rule is an empirical rule that can predict the major alkene product in an elimination reaction. It is also known Saytzeff Rule or Z-rule. Alexander Zaitsev, a Russian chemist, examined many elimination processes and discovered a common pattern in … Read more