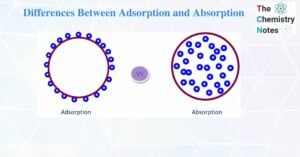
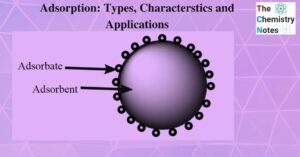
Electrolysis of Water- Definition, Principle, and Applications
The electrolysis of water produces hydrogen and oxygen gases. When hydrogen is required, this electrolytic process is used in some industrial applications. Electrolysis is the process of converting water (H2O) into hydrogen (H+) and hydroxide (OH–) ions by passing an … Read more