Solid: Properties, Classification, Types, Examples
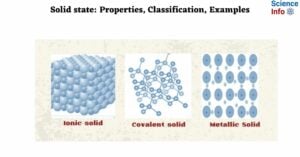
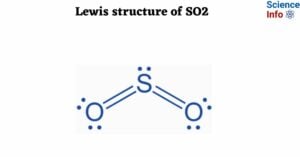
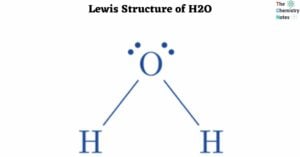
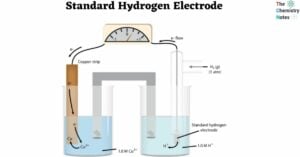
A solid is the state of matter. Solids have fixed shapes and sizes. In any compound, intermolecular forces of attraction and thermal force operate in opposite directions. The intermolecular force of attraction tends to keep the particles together while the … Read more