London Dispersion Forces: Definition, Examples, Formula
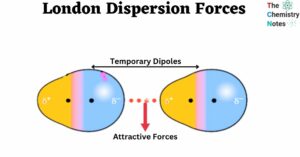
London dispersion forces are a transitory attraction between two atoms that are adjacent. The electrons of one atom are unsymmetrical, resulting in a temporary dipole. This dipole induces an induced dipole in the other atom, resulting in the attraction between … Read more