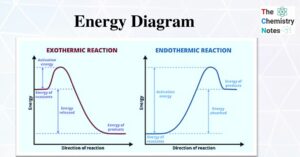
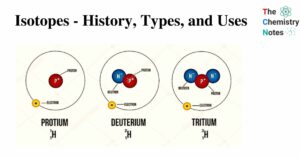
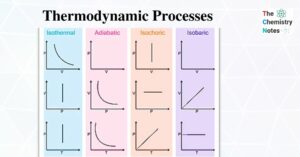
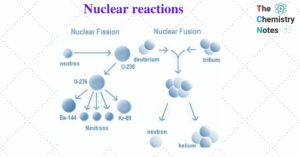
Hess’s Law- Statement, Application, and Importance
Directly determining the enthalpy change for a chemical reaction can be challenging in some cases. It might be challenging to manage the reaction that is occurring in a lab. Alternatively, taking measurements from the reaction mixture using conventional calorimetry techniques … Read more