Catalyst and Catalysis: Types, Examples, Differences
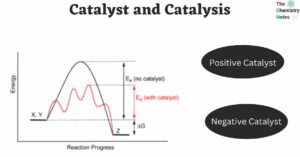
A catalyst is a substance that initiates or accelerates the rate of a particular chemical reaction without itself being chemically affected. A catalyst can be added to a reaction and then recovered and reused after the reaction occurs. The process … Read more