The diatomic elements include hydrogen(H), nitrogen(N), oxygen(O), fluorine(F), chlorine(Cl), bromine(Br), and iodine(I). In nature, the diatomic elements only exist as H2, N2, O2, F2, Cl2, Br2, and I2.
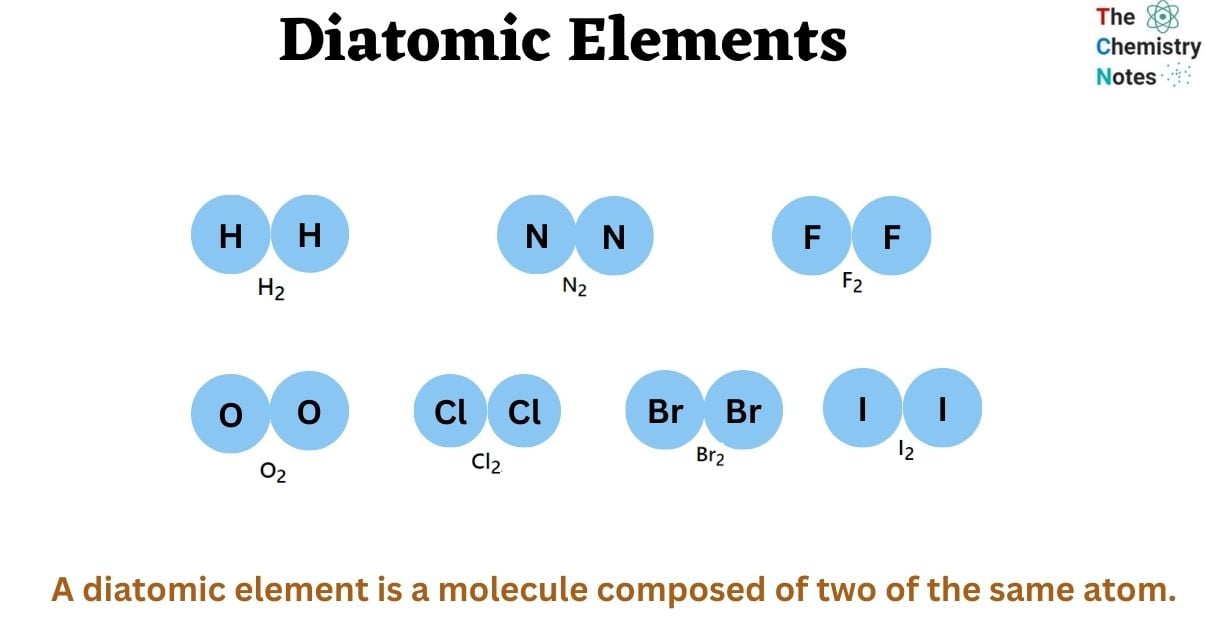
A molecule is formed when two or more atoms combine. Because the number of atoms in a molecule defines the prefix, a molecule containing two atoms is called diatomic. The prefix di-, which means “two,” is Greek in origin. A homonuclear diatomic molecule is generated by the chemical reaction of two atoms of equivalent type. For instance, a diatomic molecule is homonuclear if it contains two atoms of the same element, such as hydrogen (H2) or oxygen (O2).
Many compounds, including HCl, NaCl, and KBr, are diatomic. Similarly, when two separate atoms react or form a bond in a chemical process, they form a heteronuclear diatomic molecule. When carbon and oxygen react or form a bond, they produce a carbon monoxide molecule (C + O = CO). A nonpolar connection exists between two homonuclear diatomic molecules. Seven pure elements unite to form diatomic molecules.
Formation of Diatomic Elements
- The diatomic elements have high electronegativities. Bonding with oneself helps one become more stable.
- Five of the seven (hydrogen and the halogens) are only one electron shy of having an octet.
- They complete their octets by covalently bonding with one another.
- Nitrogen and oxygen share the same characteristics.
- Nitrogen, on the other hand, may share three of its valance electrons, leading it to triple bind with itself.
- Oxygen may create a double bond with itself by sharing two of its valence electrons.
List of Diatomic Elements
There are seven regularly occurring diatomic elements which are discussed here:
Hydrogen (H2)
- Hydrogen (H), with an atomic number of 1, is a colorless gas that was not formally identified as an element until 1766 by Henry Cavendish, but was discovered unintentionally nearly a hundred years earlier by Robert Boyle.
- As the first element in the periodic table, hydrogen is the lightest and most numerous of all the chemical elements in the universe, accounting for 75% of its mass.
- A hydrogen atom has only one valence electron in its outer layer. To become stable, the hydrogen atom need another electron to complete its layer. This is accomplished by sharing a proton with another hydrogen atom via a non-polar covalent link, resulting in a diatomic molecule.
Nitrogen (N2)
- Nitrogen (N2) has the atomic number 7. Nitrogen is the most prevalent element in the Earth’s atmosphere, accounting for approximately 78.05% of the volume of the atmosphere.
- It is an odorless, colorless, and generally inert gas, and it stays colorless and odorless in liquid form.
- A nitrogen atom has 5 valence electrons in its outermost layer. To form a stable combination, two nitrogen atoms combine and share three electrons from the opposite atom, leaving 8 electrons in the final layer. This results in a diatomic nitrogen molecule with non-polar covalent bonds between the two atoms.
Oxygen (O2)
- The element Oxygen has an atomic number of eight. This colorless, odorless gas has eight protons in its nucleus and is light blue in liquid and solid phases.
- The third most prevalent element in the universe by mass, oxygen makes about one-fifth of the atmosphere on Earth. The oxygen cycle on Earth, which is primarily fueled by plant photosynthesis, is the cause of the high oxygen content in the atmosphere.
- The outermost layer of an oxygen atom is made up of six valence electrons. It forms a non-polar covalent link with another oxygen atom in order to share two electrons, resulting in the formation of a stable diatomic molecule.
Fluorine (F2)
- Fluorine(F) has an atomic number of 9 and is the most reactive and electronegative element. This nonmetallic element is a light yellow gas that belongs to the halogen group. Fluorine is the most electronegative element in the periodic table.
- Fluorine is a very poisonous pale gas with a strong odor that occurs as a diatomic molecule at ambient temperature.
- An atom of fluorine contains 7 valence electrons in its outermost shell. By exchanging one electron with another fluorine atom through a non-polar covalent bond, it becomes stable and creates a diatomic fluorine molecule.
Chlorine (Cl2)
- Chlorine (Cl2) is an element with the atomic number 17 and is a member of the halogen group. One of its forms, NaCl, has been utilized since ancient times. At normal temperatures, chlorine appears as a yellow-green gas, however the majority of its typical compounds are colorless.
- It is composed of 3 layers of electrons, with the outermost layer containing 7 electrons. It produces a stable diatomic chlorine molecule by exchanging one electron with another chlorine atom through a non-polar covalent connection.
Bromine (Br2)
- Bromine (Br) has an atomic number of 35. It is the only non-metallic element that is a liquid at room temperature and is a viscous dark brown liquid.
- When the temperature rises and this element becomes gaseous, it produces diatomic molecules. The outermost layer has 7 electrons and requires 1 to become stable and create Br2.
Iodine (I2)
- Iodine (I) has an atomic number of 53 and a nucleus of 53 protons. At room temperature, iodine is a purple-black nonmetallic solid which is particularly essential in organic chemistry.
- Iodine is essential in the biology of all living beings since its lack causes disorders such as hyperthyroidism and hypothyroidism.
- It transforms into a diatomic I2 molecule when heated, just as bromine. Iodine atoms exchange electrons with each other to complete their octets and achieve stability.
These elements are nonmetals because halogens are a type of nonmetallic element. Bromine is a liquid at ambient temperature, whereas the other elements are all gases under normal circumstances. When the temperature or pressure is reduced, the other elements become diatomic liquids.
While only these seven elements usually form diatomic molecules, additional elements can as well. However, diatomic molecules generated by other elements are not particularly stable, and their bonds are rapidly broken.
Physical Properties of Diatomic Elements
- At room temperature, five of the seven diatomic elements are gases.
- The physical characteristics of covalently bound molecules are caused by intermolecular forces.
- Iodine and bromine are the only two exceptions. Iodine is a solid, while bromine is a liquid.
- The diatomic elements are exclusively subject to Van der Waal forces.
- Bromine and iodine both have enough electrons to form powerful temporary dipoles.
- The London Dispersion Effect causes temporary dipoles.
Chemical Properties of Diatomic Elements
- Diatomic elements are employed in a variety of operations.
- Bond length increases as we move down the periodic table.
- Since Br2 and I2 have large bond lengths, they have lower bond energy. As a result, B r 2 and I 2 are more reactive than the other diatomic elements.
- A few reactions, in general, are energetically advantageous and occur spontaneously.
- However diatomic atoms are highly stable, chemists usually add a catalyst to lower the activation energy.
Video Reference
References
- https://www.vedantu.com/question-answer/are-the-eight-diatomic-elements-class-11-chemistry-cbse-60dac3e6b92fd7057b0ab445
- https://chem.libretexts.org/Courses/Portland_Community_College/CH151%3A_Preparatory_Chemistry/05%3A_The_Nuclei_of_Atoms/5.02%3A_Monotomic_and_Diatomic_Elements
- https://sciencenotes.org/diatomic-elements/
- https://www.chemicool.com/definition/diatomic.html
- https://www.scienceabc.com/pure-sciences/what-are-diatomic-molecules.html
- https://www.expii.com/t/diatomic-elements-importance-examples-11149
- https://warbletoncouncil.org/elementos-diatomicos-6943
