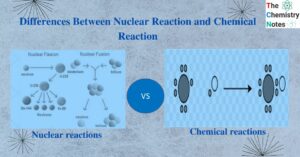
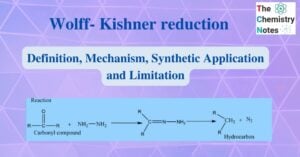
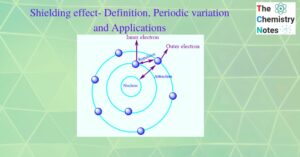
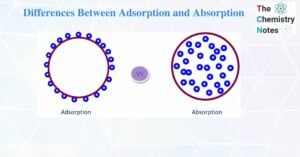
Differences between lyophilic and lyophobic sols
A colloid is a heterogenous mixture in which particle size ranges between 1 and 100 nm. The colloidal state consists of two phases: a dispersed phase and a dispersion phase. Dispersed phase: The substance is distributed as colloidal particles. Dispersed … Read more