Formal Charge Calculation
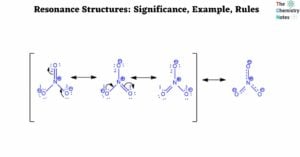
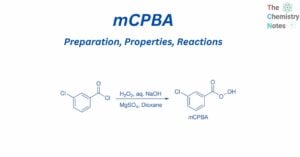
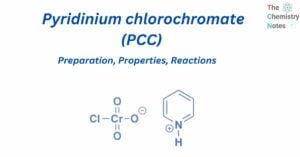
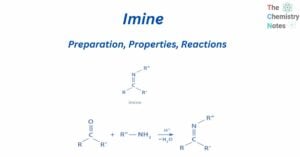
Formal charge calculation is significant since it is used to determine the lowest energy configuration among the different potential Lewis structures of a molecule. An atom in a molecule is given a formal charge if all of the electrons in the … Read more