Lewis Structure: Definition, Important, Limitations
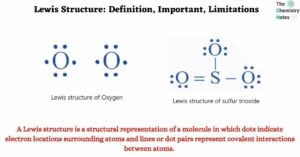
A Lewis structure is a diagram that depicts the chemical bonds that exist between atoms in a molecule and their valence electrons, also known as lone pairs of electrons. The diagram is also known as a Lewis dot diagram, a … Read more