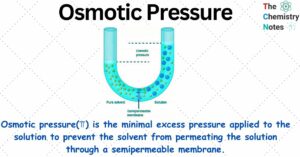
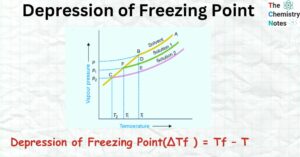
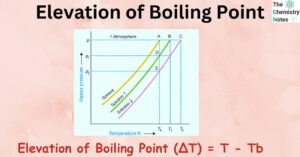
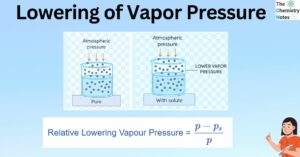
Mixture: 2 Important Types, Properties
A mixture is made up of one or more pure components with different ratios. Mixtures can be either heterogeneous or homogeneous. When compared to homogeneous mixtures, heterogeneous mixtures can be visually divided into individual components. The most prevalent kind of … Read more