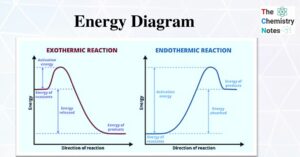
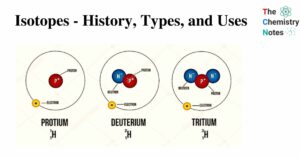
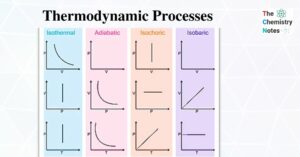
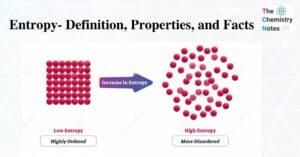
Thermodynamics and Laws of Thermodynamics
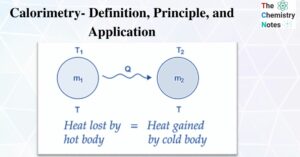
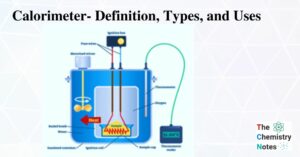
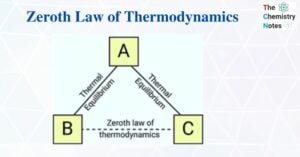
Thermodynamics is the branch of physics that studies the relationship between heat and other properties of a substance (such as pressure, density, temperature, and so on). Thermodynamics, in particular, focuses on how heat transfer is related to various energy changes … Read more